Is Research on "Synthetic Cells" Moving to the Next Level?
- PMID: 30587790
- PMCID: PMC6463193
- DOI: 10.3390/life9010003
Is Research on "Synthetic Cells" Moving to the Next Level?
Abstract
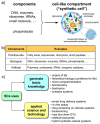
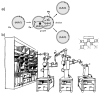
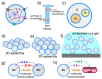
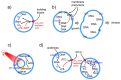
"Synthetic cells" research focuses on the construction of cell-like models by using solute-filled artificial microcompartments with a biomimetic structure. In recent years this bottom-up synthetic biology area has considerably progressed, and the field is currently experiencing a rapid expansion. Here we summarize some technical and theoretical aspects of synthetic cells based on gene expression and other enzymatic reactions inside liposomes, and comment on the most recent trends. Such a tour will be an occasion for asking whether times are ripe for a sort of qualitative jump toward novel SC prototypes: is research on "synthetic cells" moving to a next level?
Keywords: artificial cells; autopoiesis; cell-free protein synthesis; complexity; liposomes; microfluidics; numerical modeling; origins of life; protocells; synthetic biology; synthetic cells.
Conflict of interest statement
The author declares no conflict of interest.
Figures




References
LinkOut - more resources
Full Text Sources

