A personalized and long-acting local therapeutic platform combining photothermal therapy and chemotherapy for the treatment of multidrug-resistant colon tumor
- PMID: 30587968
- PMCID: PMC6294077
- DOI: 10.2147/IJN.S184728
A personalized and long-acting local therapeutic platform combining photothermal therapy and chemotherapy for the treatment of multidrug-resistant colon tumor
Abstract
Background: Local photothermal therapy (PTT) provides an easily applicable, noninvasive adjunctive therapy for colorectal cancer (CRC), especially when multidrug resistance (MDR) occurs. However, using PTT alone does not result in complete tumor ablation in many cases, thus resulting in tumor recurrence and metastasis.
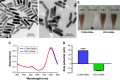
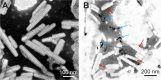
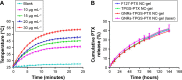
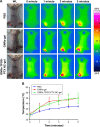
Materials and methods: In this study, we aim to develop a personalized local therapeutic platform combining PTT with long-acting chemotherapy for the treatment of MDR CRC. The platform consists of polyethylene glycol (PEG)-coated gold nanorods (PEG-GNRs) and D-alpha-tocopheryl PEG 1000 succinate (TPGS)-coated paclitaxel (PTX) nanocrystals (TPGS-PTX NC), followed by the incorporation into an in situ hydrogel (gel) system (GNRs-TPGS-PTX NC-gel) before injection. After administration, PEG-GNRs can exert quick and efficient local photothermal response under near-infrared laser irradiation to shrink tumor; TPGS-PTX NC then provides a long-acting chemotherapy due to the sustained release of PTX along with the P-glycoprotein inhibitor TPGS to reverse the drug resistance.
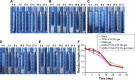
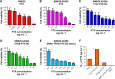
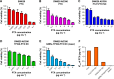
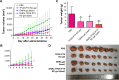
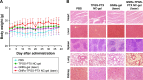
Results: The cytotoxicity studies showed that the IC50 of GNRs-TPGS-PTX NC-gel with laser irradiation decreased to ~178-folds compared with PTX alone in drug-resistant SW620 AD300 cells. In the in vivo efficacy test, after laser irradiation, the GNRs-TPGS-PTX NC-gel showed similar tumor volume inhibition compared with GNRs-gel at the beginning. However, after 14 days, the tumor volume of the mice treated with GNRs-gel quickly increased, while that of the mice treated with GNRs-TPGS-PTX NC-gel remained controllable due to the long-term chemotherapeutic effect of TPGS-PTX NC. The mice treated with GNRs-TPGS-PTX NC-gel also showed no weight loss and obvious organ damages and lesions during the treatment, indicating a low systemic side effect profile and a good biocompatibility.
Conclusion: Overall, the nano-complex may serve as a promising local therapeutic patch against MDR CRC with one-time dosing to achieve a long-term tumor control. The doses of PEG-GNRs and TPGS-PTX NC can be easily adjusted before use according to patient-specific characteristics potentially making it a personalized therapeutic platform.
Keywords: TPGS; gold nanorods; in situ hydrogel; paclitaxel nanocrystals; tumor recurrence.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures


Similar articles
-
A multi-functionalized nanocomposite constructed by gold nanorod core with triple-layer coating to combat multidrug resistant colorectal cancer.Mater Sci Eng C Mater Biol Appl. 2020 Feb;107:110224. doi: 10.1016/j.msec.2019.110224. Epub 2019 Oct 14. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 31761194
-
D-α-tocopherol polyethylene glycol succinate-based redox-sensitive paclitaxel prodrug for overcoming multidrug resistance in cancer cells.Mol Pharm. 2014 Sep 2;11(9):3196-209. doi: 10.1021/mp500384d. Epub 2014 Aug 14. Mol Pharm. 2014. PMID: 25102234
-
Biodegradable mixed MPEG-SS-2SA/TPGS micelles for triggered intracellular release of paclitaxel and reversing multidrug resistance.Int J Nanomedicine. 2016 Oct 6;11:5109-5123. doi: 10.2147/IJN.S111930. eCollection 2016. Int J Nanomedicine. 2016. PMID: 27785018 Free PMC article.
-
Recent developments in d-α-tocopheryl polyethylene glycol-succinate-based nanomedicine for cancer therapy.Drug Deliv. 2017 Nov;24(1):1831-1842. doi: 10.1080/10717544.2017.1406561. Drug Deliv. 2017. PMID: 29182031 Free PMC article. Review.
-
Recent Advances in the Application of Vitamin E TPGS for Drug Delivery.Theranostics. 2018 Jan 1;8(2):464-485. doi: 10.7150/thno.22711. eCollection 2018. Theranostics. 2018. PMID: 29290821 Free PMC article. Review.
Cited by
-
Gold as a Possible Alternative to Platinum-Based Chemotherapy for Colon Cancer Treatment.Cancers (Basel). 2019 Jun 5;11(6):780. doi: 10.3390/cancers11060780. Cancers (Basel). 2019. PMID: 31195711 Free PMC article. Review.
-
Multifunctional Gold Nanoparticles in Cancer Diagnosis and Treatment.Int J Nanomedicine. 2022 May 6;17:2041-2067. doi: 10.2147/IJN.S355142. eCollection 2022. Int J Nanomedicine. 2022. PMID: 35571258 Free PMC article. Review.
-
Nanomedicine to Overcome Multidrug Resistance Mechanisms in Colon and Pancreatic Cancer: Recent Progress.Cancers (Basel). 2021 Apr 24;13(9):2058. doi: 10.3390/cancers13092058. Cancers (Basel). 2021. PMID: 33923200 Free PMC article. Review.
-
The Controlled Release and Prevention of Abdominal Adhesion of Tannic Acid and Mitomycin C-Loaded Thermosensitive Gel.Polymers (Basel). 2023 Feb 16;15(4):975. doi: 10.3390/polym15040975. Polymers (Basel). 2023. PMID: 36850258 Free PMC article.
-
Novel strategies to reverse chemoresistance in colorectal cancer.Cancer Med. 2023 May;12(10):11073-11096. doi: 10.1002/cam4.5594. Epub 2023 Jan 16. Cancer Med. 2023. PMID: 36645225 Free PMC article. Review.
References
-
- Siegel RL, Miller KD, Fedewa SA, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67(3):177–193. - PubMed
-
- Saltzman WM, Fung LK. Polymeric implants for cancer chemotherapy. Adv Drug Deliv Rev. 1997;26(2–3):209–230. - PubMed
-
- Shannon AM, Bouchier-Hayes DJ, Condron CM, Toomey D. Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Cancer Treat Rev. 2003;29(4):297–307. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

