Drug delivery to atherosclerotic plaques using superparamagnetic iron oxide nanoparticles
- PMID: 30587970
- PMCID: PMC6294059
- DOI: 10.2147/IJN.S179273
Drug delivery to atherosclerotic plaques using superparamagnetic iron oxide nanoparticles
Abstract
Introduction: Magnetic drug targeting utilizes superparamagnetic iron oxide nanoparticles (SPIONs) to accumulate drugs in specified vasculature regions.
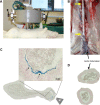
Methods: We produced SPIONs conjugated with dexamethasone phosphate (SPION-DEXA). The efficacy of magnetic drug targeting was investigated in a rabbit model of atherosclerosis induced by balloon injury and high cholesterol diet.
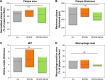
Results: In vitro, SPION-DEXA were well-tolerated by endothelial cells. SPION-DEXA were internalized by human peripheral blood mononuclear cells and induced CD163 expression comparable with the free drug. In vivo, magnetic targeting of SPIONs to abdominal aorta was confirmed by histology. Upon vascular injury followed by high-cholesterol diet, early administration of SPION-DEXA enhanced the inflammatory burden in the plaques. Increased macrophage content and larger intima- media thickness were observed in animals treated with SPION-DEXA compared with controls. In advanced atherosclerosis, no beneficial effect of local glucocorticoid therapy was detectable.
Conclusion: Magnetic drug targeting represents an efficient platform to deliver drugs to diseased arteries in vivo. However, targeting of vascular injury in the lipid-rich environment using dexamethasone-conjugated SPIONs may cause accelerated inflammatory response.
Keywords: dexamethasone; macrophage accumulation; magnetic drug targeting; magnetic nanoparticles; rabbit model of atherosclerosis.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures




References
-
- Libby P, Theroux P. Pathophysiology of coronary artery disease. Circulation. 2005;111(25):3481–3488. - PubMed
-
- Cicha I, Garlichs CD, Alexiou C. Cardiovascular therapy through nanotechnology – how far are we still from bedside? Eur J Nanomed. 2014;6(2):63–87.
-
- Thorat ND, Bohara RA, Yadav HM, Tofail SAM. Multi-modal MR imaging and magnetic hyperthermia study of Gd doped Fe3O4 nanoparticles for integrative cancer therapy. RSC Adv. 2016;6(97):94967–94975.
-
- Lyer S, Tietze R, Jurgons R, et al. Visualisation of tumour regression after local chemotherapy with magnetic nanoparticles – a pilot study. Anticancer Res. 2010;30(5):1553–1557. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

