Synaptic Reorganization Response in the Cochlear Nucleus Following Intense Noise Exposure
- PMID: 30593923
- PMCID: PMC6595490
- DOI: 10.1016/j.neuroscience.2018.12.023
Synaptic Reorganization Response in the Cochlear Nucleus Following Intense Noise Exposure
Abstract
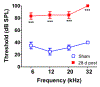
The cochlear nucleus, located in the brainstem, receives its afferent auditory input exclusively from the auditory nerve fibers of the ipsilateral cochlea. Noise-induced neurodegenerative changes occurring in the auditory nerve stimulate a cascade of neuroplastic changes in the cochlear nucleus resulting in major changes in synaptic structure and function. To identify some of the key molecular mechanisms mediating this synaptic reorganization, we unilaterally exposed rats to a high-intensity noise that caused significant hearing loss and then measured the resulting changes in a synaptic plasticity gene array targeting neurogenesis and synaptic reorganization. We compared the gene expression patterns in the dorsal cochlear nucleus (DCN) and ventral cochlear nucleus (VCN) on the noise-exposed side versus the unexposed side using a PCR gene array at 2 d (early) and 28 d (late) post-exposure. We discovered a number of differentially expressed genes, particularly those related to synaptogenesis and regeneration. Significant gene expression changes occurred more frequently in the VCN than the DCN and more changes were seen at 28 d versus 2 d post-exposure. We confirmed the PCR findings by in situ hybridization for Brain-derived neurotrophic factor (Bdnf), Homer-1, as well as the glutamate NMDA receptor Grin1, all involved in neurogenesis and plasticity. These results suggest that Bdnf, Homer-1 and Grin1 play important roles in synaptic remodeling and homeostasis in the cochlear nucleus following severe noise-induced afferent degeneration.
Keywords: dorsal cochlear nucleus; gene expression; noise-induced hearing loss; synaptic plasticity; ventral cochlear nucleus.
Copyright © 2018 IBRO. Published by Elsevier Ltd. All rights reserved.
Figures

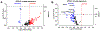
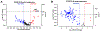





References
-
- Bashir ZI, Bortolotto ZA, Davies CH, Berretta N, Irving AJ, Seal AJ, Henley JM, Jane DE, Watkins JC, Collingridge GL (1993) Induction of LTP in the hippocampus needs synaptic activation of glutamate metabotropic receptors. Nature 363:347–350. - PubMed
-
- Becker E, Soler RM, Yuste VJ, Gine E, Sanz-Rodriguez C, Egea J, Martin-Zanca D, Comella JX (1998) Development of survival responsiveness to brain-derived neurotrophic factor, neurotrophin 3 and neurotrophin 4/5, but not to nerve growth factor, in cultured motoneurons from chick embryo spinal cord [In Process Citation] J Neurosci 18:7903–7911. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

