Oncologic Outcomes in Metastatic Colorectal Cancer with Regorafenib with FOLFIRI as a Third- or Fourth-Line Setting
- PMID: 30594039
- PMCID: PMC6307535
- DOI: 10.1016/j.tranon.2018.12.003
Oncologic Outcomes in Metastatic Colorectal Cancer with Regorafenib with FOLFIRI as a Third- or Fourth-Line Setting
Abstract
Background: To evaluate the efficacy and toxicities of regorafenib plus irinotecan, dose-escalated on the basis of uridine diphosphate glucuronosyltransferase 1A1 (UGT1A1) genotyping, in previously heavily treated metastatic colorectal cancer (mCRC) and the prognostic values of EGFR expression, KRAS mutations, and tumor sidedness.
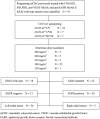
Methods: Forty-one patients with mCRC with disease progression after treatment with fluoropyrimidines, oxaliplatin, irinotecan, anti-VEGF, and anti-EGFR MoAbs were subjected to UGT1A1 genotyping and received regorafenib combined with FOLFIRI with dose-escalated irinotecan.
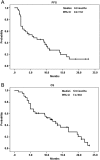
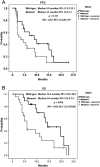
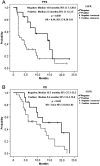
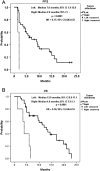
Results: The median follow-up period was 10.0 months (1.3-23.5 months). The overall disease control rate was 58.5%, whereas the median progression-free survival (PFS) and overall survival (OS) were 6.0 months and 12.0 months, respectively. KRAS mutations were significantly associated with positive EGFR expression (P = .026). KRAS mutations significantly correlated with a shorter OS than KRAS wild-type (6.0 vs. 14.4 months, P = .014) but had no significant association with PFS. Positive EGFR expression had an inverse correlation with PFS (2.5 vs. 14.0 months, P = .039) and OS (9.6 vs. 19.7 months, P = .044). Moreover, left-sided tumors associated with superior PFS (2.0 vs. 7.0 months, P < .0001) and OS (4.0 vs. 13.0 months, P < .0001), and tumor sidedness was an independent prognostic factor by the multivariate analysis.
Conclusion: Regorafenib and FOLFIRI concomitant therapy with dose-escalated irinotecan seemed to be potentially practicable with satisfactory oncological results. KRAS mutations and EGFR expression might be predictors of poor oncological outcomes; however, left-sided mCRCs would be more beneficial for concomitant regorafenib and FOLFIRI therapy.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, Comber H, Forman D, Bray F. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–1403. - PubMed
-
- Arnold D, Stein A. New developments in the second-line treatment of metastatic colorectal cancer: potential place in therapy. Drugs. 2013;73:883–891. - PubMed
-
- Wilhelm SM, Dumas J, Adnane L, Lynch M, Carter CA, Schutz G, Thierauch KH, Zopf D. Regorafenib (BAY 73-4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer. 2011;129:245–255. - PubMed
-
- Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouche O, Mineur L, Barone C. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:303–312. - PubMed
-
- Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, Xu J, Bai Y, Chi Y, Wang L. Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2015;16:619–629. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

