Adipose stem cell crosstalk with chemo-residual breast cancer cells: implications for tumor recurrence
- PMID: 30594967
- PMCID: PMC6422973
- DOI: 10.1007/s10549-018-05103-w
Adipose stem cell crosstalk with chemo-residual breast cancer cells: implications for tumor recurrence
Abstract
Purpose: Most triple-negative breast cancer (TNBC) patients exhibit an incomplete response to neoadjuvant chemotherapy, resulting in chemo-residual tumor cells that drive tumor recurrence and patient mortality. Accordingly, strategies for eliminating chemo-residual tumor cells are urgently needed. Although stromal cells contribute to tumor cell invasion, to date, their ability to influence chemo-residual tumor cell behavior has not been examined. Our study is the first to investigate cross-talk between adipose-derived stem cells (ASCs) and chemo-residual TNBC cells. We examine if ASCs promote chemo-residual tumor cell proliferation, having implications for tumor recurrence.
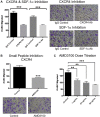
Methods: ASC migration toward chemo-residual TNBC cells was tested in a transwell migration assay. Importance of the SDF-1α/CXCR4 axis was determined using neutralizing antibodies and a small molecule inhibitor. The ability of ASCs to drive tumor cell proliferation was analyzed by culturing tumor cells ± ASC conditioned media (CM) and determining cell counts. Downstream signaling pathways activated in chemo-residual tumor cells following their exposure to ASC CM were studied by immunoblotting. Importance of FGF2 in promoting proliferation was assessed using an FGF2-neutralizing antibody.
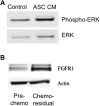
Results: ASCs migrated toward chemo-residual TNBC cells in a CXCR4/SDF-1α-dependent manner. Moreover, ASC CM increased chemo-residual tumor cell proliferation and activity of extracellular signal-regulated kinase (ERK). An FGF2-neutralizing antibody inhibited ASC-induced chemo-residual tumor cell proliferation.
Conclusions: ASCs migrate toward chemo-residual TNBC cells via SDF-1α/CXCR4 signaling, and drive chemo-residual tumor cell proliferation in a paracrine manner by secreting FGF2 and activating ERK. This paracrine signaling can potentially be targeted to prevent tumor recurrence.
Keywords: Adipose-derived stem cells (ASCs); Fibroblast growth factor 2 (FGF2); Migration; Proliferation; Recurrence; Triple-negative breast cancer (TNBC).
Conflict of interest statement
Conflict of interest
No authors on this manuscript declare a conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Figures



References
-
- Rouzier R, Perou CM, Symmans WF, Ibrahim N, Cristofanilli M, Anderson K, Hess KR, Stec J, Ayers M, Wagner P, Morandi P, Fan C, Rabiul I, Ross JS, Hortobagyi GN, Pusztai L. Breast cancer molecular subtypes respond differently to preoperative chemotherapy. Clinical Cancer Res. 2005;11(16):5678–5685. doi: 10.1158/1078-0432.CCR-04-2421. - DOI - PubMed
-
- Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B, Green M, Cristofanilli M, Hortobagyi GN, Pusztai L. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26(8):1275–1281. doi: 10.1200/JCO.2007.14.4147. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

