Device implantation for patients on antiplatelets and anticoagulants: Use of suction drain
- PMID: 30595295
- PMCID: PMC6309121
- DOI: 10.1016/j.ihj.2017.12.009
Device implantation for patients on antiplatelets and anticoagulants: Use of suction drain
Abstract
Background and objectives: Cardiovascular implantable electronic devices (CIED) are frequently implanted in patients on anti-thrombotic agents. Pocket hematomas are more likely to occur in these patients. The use of a sterile surgical drain in the pulse generator pocket site could prevent hematomas, but fear of infection precludes its use. The objective of the present study is to study the safety and efficacy of surgical drain in patients on antithrombotics undergoing CIED implantations.
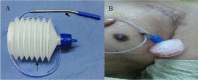
Methods: This is a single-centre, retrospective study involving patients undergoing CIED implantations on antithrombotics (antiplatelets and anticoagulants) from August 2013 to July 2016. Patients with high risk of thromboembolism were continued on oral antithrombotics or were bridged with heparin after stopping oral antithrombotics. A sterile close wound suction drain was placed in device pockets following CIED implantations. Post procedure, pressure dressing was applied and removed after 12 h once the drain volume was less than 10 ml in 24 h.
Results: Sixty seven patients required surgical drain implantation. Major indications for antithrombotic use were presence of intracoronary stent, atrial fibrillation and mechanical valve replacements. The mean post-procedural hospital stay was 3 ± 0.9 days and mean overall drain was 16.6 ± 8.2 ml. At a mean follow up of 17.6 ± 8.2 months, one patient (1.4%) had pocket hematoma. There were no infections.
Conclusion: The use of a surgical drain in CIED implantation significantly reduces the risk of hematoma formation without increasing the risk of infection. Antithrombotic drugs can be safely continued at the time of implantation of cardiac devices.
Keywords: Anticoagulation; Antiplatelets; Antithrombotic treatment; Device implantation; Pocket hematoma; Surgical drain.
Copyright © 2017. Published by Elsevier B.V.
Figures
Similar articles
-
Current strategies to minimize postoperative hematoma formation in patients undergoing cardiac implantable electronic device implantation: A review.Heart Rhythm. 2021 Apr;18(4):641-650. doi: 10.1016/j.hrthm.2020.11.017. Epub 2020 Nov 23. Heart Rhythm. 2021. PMID: 33242669 Review.
-
Pocket related complications following cardiac electronic device implantation in patients receiving anticoagulation and/or dual antiplatelet therapy: prospective evaluation of different preventive strategies.J Interv Card Electrophysiol. 2019 Apr;54(3):247-255. doi: 10.1007/s10840-018-0488-y. Epub 2018 Nov 21. J Interv Card Electrophysiol. 2019. PMID: 30460588
-
Risk factors associated with bleeding after multi antithrombotic therapy during implantation of cardiac implantable electronic devices.Heart Vessels. 2017 Mar;32(3):333-340. doi: 10.1007/s00380-016-0879-x. Epub 2016 Jul 28. Heart Vessels. 2017. PMID: 27469320 Free PMC article.
-
Pocket haematoma after cardiac electronic device implantation in patients receiving antiplatelet and anticoagulant treatment: a single-centre experience.Acta Cardiol. 2017 Feb;72(1):47-52. doi: 10.1080/00015385.2017.1281539. Acta Cardiol. 2017. PMID: 28597740
-
Management of antithrombotic therapy in patients undergoing electrophysiological device surgery.Europace. 2015 Jun;17(6):840-54. doi: 10.1093/europace/euu357. Epub 2015 Feb 24. Europace. 2015. PMID: 25712980 Review.
Cited by
-
EHRA expert consensus statement and practical guide on optimal implantation technique for conventional pacemakers and implantable cardioverter-defibrillators: endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), and the Latin-American Heart Rhythm Society (LAHRS).Europace. 2021 Jul 18;23(7):983-1008. doi: 10.1093/europace/euaa367. Europace. 2021. PMID: 33878762 Free PMC article.
References
-
- Mond H.G., Proclemer A. The 11th world survey of cardiac pacing and arrhythmia’s project. Pacing Clin Electrophysiol. 2011;34(8):1013–1027. - PubMed
-
- Ector H., P.V Current use of pacemakers, implantable cardioverter defibrillators, and resynchronization devices: data from the registry of the European Heart Rhythm Association. Eur Hear J Suppl. 2007;9:144–149.
-
- Bardy G.H., Lee K.L., Mark D.B. Amiodarone or an implantable cardioverter–defibrillator for congestive heart failure. N Engl J Med. 2005;352(3):225–237. - PubMed
-
- Greenspon Arnold J., Hart Robert G., Dawson David. Predictors of stroke in patients paced for sick sinus syndrome. J Am Coll Cardiol. 2004;43(9):1617–1622. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical