Moxibustion for prehypertension and stage I hypertension: a pilot randomized controlled trial
- PMID: 30596012
- PMCID: PMC6309023
- DOI: 10.1016/j.imr.2018.11.002
Moxibustion for prehypertension and stage I hypertension: a pilot randomized controlled trial
Abstract
Background: Prehypertension and hypertension are associated with cardiovascular disease, ischemic heart disease, and stroke morbidity. The purpose of this study is to evaluate the effectiveness and safety of moxibustion in patients with prehypertension or hypertension.
Methods: Forty-five subjects with prehypertension or stage I hypertension were randomized into three groups: moxibustion treatment group A (2 sessions/week for 4 weeks), moxibustion treatment group B (3 sessions/week for 4 weeks), and control group (nontreated group). The primary outcome measure was the change in blood pressure after 4 weeks of treatment. Safety was assessed at every visit.
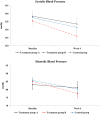
Results: There were no significant differences in systolic blood pressure (SBP) or diastolic blood pressure (DBP) among three groups after 4 weeks of treatment (p = 0.4798 and p = 0.3252, respectively). In treatment group B, there was a significant decrease in SBP and DBP from baseline to 4 weeks of treatment (mean difference (MD) -9.55; p = 0.0225, MD -7.55; p = 0.0098, respectively). There were no significant differences among groups in secondary outcome measures after 4 weeks of treatment. Six adverse events (AEs) in the treatment group A and 12 AEs in the treatment group B occurred related to the moxibustion treatment.
Conclusion: In conclusion, the results of this study show that moxibustion (3 sessions/week for 4 weeks) might lower blood pressure in patients with prehypertension or stage I hypertension and treatment frequency might affect effectiveness of moxibustion in BP regulation. Further randomized controlled trials with a large sample size on prehypertension and hypertension should be conducted.
Trial registration: This study was registered with the 'Clinical Research Information Service (CRIS)', Republic of Korea (KCT0000469), and the protocol for this study was presented orally at the 15th International Council of Medical Acupuncture and Related Techniques (ICMART) in Athens, 25-27 May 2012.
Keywords: AE, adverse event; BMI, body mass index; BP, blood pressure; CI, confidence interval; DBP, diastolic blood pressure; EQ-5D, EuroQol-5 Dimensions; FSS, Fatigue Severity Scale; HRV, heart rate variability; Hypertension; MD, mean difference; Moxibustion; NDI, neck disability index; PSQI, Pittsburgh Sleep Quality Index; Prehypertension; RCT, randomized controlled trial; SAE, serious adverse event; SBP, systolic blood pressure; SRI-MF, Modified Form of the Stress Response Inventory.
Figures
References
-
- Castelli W. Epidemiology of coronary heart disease: the Framingham study. Am J Med. 1984;76:4–12. - PubMed
-
- Organization WH, Group ISoHW 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J Hypert. 2003;21:1983–1992. - PubMed
-
- Kearney P.M., Whelton M., Reynolds K., Muntner P., Whelton P.K., He J. Global burden of hypertension: analysis of worldwide data. Lancet. 2005;365:217–223. - PubMed
-
- Chobanian A.V., Bakris G.L., Black H.R., Cushman W.C., Green L.A., Izzo J.L. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension. 2003;42:1206–1252. - PubMed
-
- Choi K.M., Park H.S., Han J.H., Lee J.S., Lee J., Ryu O.H. Prevalence of prehypertension and hypertension in a Korean population: Korean National Health and Nutrition Survey 2001. J Hypert. 2006;24:1515–1521. - PubMed
LinkOut - more resources
Full Text Sources