Thermodynamics of voltage-gated ion channels
- PMID: 30596139
- PMCID: PMC6276078
- DOI: 10.1007/s41048-018-0074-y
Thermodynamics of voltage-gated ion channels
Abstract
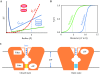
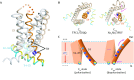
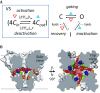
Ion channels are essential for cellular signaling. Voltage-gated ion channels (VGICs) are the largest and most extensively studied superfamily of ion channels. They possess modular structural features such as voltage-sensing domains that encircle and form mechanical connections with the pore-forming domains. Such features are intimately related to their function in sensing and responding to changes in the membrane potential. In the present work, we discuss the thermodynamic mechanisms of the VGIC superfamily, including the two-state gating mechanism, sliding-rocking mechanism of the voltage sensor, subunit cooperation, lipid-infiltration mechanism of inactivation, and the relationship with their structural features.
Keywords: Gating mechanism; Inactivation; Ion channel; Membrane potential; Sliding-rocking model; Voltage sensor.
Conflict of interest statement
Xuejun C. Zhang, Hanting Yang, Zhenfeng Liu, and Fei Sun declare that they have no conflict of interest.This article does not contain any studies with human or animal subjects performed by any of the authors.
Figures

Similar articles
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Modulation of voltage-gated ion channels by sialylation.Compr Physiol. 2012 Apr;2(2):1269-301. doi: 10.1002/cphy.c110044. Compr Physiol. 2012. PMID: 23798301 Review.
-
Bipolar switching by HCN voltage sensor underlies hyperpolarization activation.Proc Natl Acad Sci U S A. 2019 Jan 8;116(2):670-678. doi: 10.1073/pnas.1816724116. Epub 2018 Dec 26. Proc Natl Acad Sci U S A. 2019. PMID: 30587580 Free PMC article.
-
A complicated complex: Ion channels, voltage sensing, cell membranes and peptide inhibitors.Neurosci Lett. 2018 Jul 13;679:35-47. doi: 10.1016/j.neulet.2018.04.030. Epub 2018 Apr 21. Neurosci Lett. 2018. PMID: 29684532 Review.
-
Molecular basis of the interaction between gating modifier spider toxins and the voltage sensor of voltage-gated ion channels.Sci Rep. 2016 Sep 28;6:34333. doi: 10.1038/srep34333. Sci Rep. 2016. PMID: 27677715 Free PMC article.
Cited by
-
Two-state model explaining thermodynamic regulation of thermo-gating channels.Biophys Rep. 2022 Aug 31;8(4):205-211. doi: 10.52601/bpr.2022.220012. Biophys Rep. 2022. PMID: 37288006 Free PMC article.
-
Proton Quantum Tunneling: Influence and Relevance to Acidosis-Induced Cardiac Arrhythmias/Cardiac Arrest.Pathophysiology. 2021 Sep 3;28(3):400-436. doi: 10.3390/pathophysiology28030027. Pathophysiology. 2021. PMID: 35366283 Free PMC article.
-
Interplay between the electrostatic membrane potential and conformational changes in membrane proteins.Protein Sci. 2019 Mar;28(3):502-512. doi: 10.1002/pro.3563. Epub 2019 Jan 10. Protein Sci. 2019. PMID: 30549351 Free PMC article. Review.
-
The Quantum Tunneling of Ions Model Can Explain the Pathophysiology of Tinnitus.Brain Sci. 2022 Mar 23;12(4):426. doi: 10.3390/brainsci12040426. Brain Sci. 2022. PMID: 35447958 Free PMC article.
-
Molecular Insights Into the Gating Kinetics of the Cardiac hERG Channel, Illuminated by Structure and Molecular Dynamics.Front Pharmacol. 2021 Jun 8;12:687007. doi: 10.3389/fphar.2021.687007. eCollection 2021. Front Pharmacol. 2021. PMID: 34168566 Free PMC article. Review.
References
-
- Anishkin A, Akitake B, Kamaraju K, Chiang CS, Sukharev S. Hydration properties of mechanosensitive channel pores define the energetics of gating. J Phys. 2010;22:454120. - PubMed
-
- Armstrong CM, Bezanilla F. Currents related to movement of the gating particles of the sodium channels. Nature. 1973;242:459–461. - PubMed
LinkOut - more resources
Full Text Sources
