Structural Basis of Partial Agonism at the β2-Adrenergic Receptor
- PMID: 30596501
- PMCID: PMC7115863
- DOI: 10.1021/acs.biochem.8b01237
Structural Basis of Partial Agonism at the β2-Adrenergic Receptor
Conflict of interest statement
The author declares no competing financial interest.
Figures
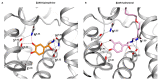
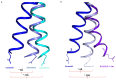
Comment on
-
Structural insights into binding specificity, efficacy and bias of a β2AR partial agonist.Nat Chem Biol. 2018 Nov;14(11):1059-1066. doi: 10.1038/s41589-018-0145-x. Epub 2018 Oct 16. Nat Chem Biol. 2018. PMID: 30327561 Free PMC article.
Similar articles
-
Stimulation of spinal dorsal horn β2-adrenergic receptor ameliorates neuropathic mechanical hypersensitivity through a reduction of phosphorylation of microglial p38 MAP kinase and astrocytic c-jun N-terminal kinase.Neurochem Int. 2016 Dec;101:144-155. doi: 10.1016/j.neuint.2016.11.004. Epub 2016 Nov 10. Neurochem Int. 2016. PMID: 27840124
-
Unraveling the Structural Basis of Biased Agonism in the β2-Adrenergic Receptor Through Molecular Dynamics Simulations.Proteins. 2025 Mar;93(3):728-744. doi: 10.1002/prot.26766. Epub 2024 Nov 16. Proteins. 2025. PMID: 39548888
-
Tyrosine 308 is necessary for ligand-directed Gs protein-biased signaling of β2-adrenoceptor.J Biol Chem. 2014 Jul 11;289(28):19351-63. doi: 10.1074/jbc.M114.558882. Epub 2014 May 15. J Biol Chem. 2014. PMID: 24831005 Free PMC article.
-
Molecular and genetic basis of beta2-adrenergic receptor function.J Allergy Clin Immunol. 1999 Aug;104(2 Pt 2):S42-6. doi: 10.1016/s0091-6749(99)70272-1. J Allergy Clin Immunol. 1999. PMID: 10452787 Review.
-
Potential Signal Transduction Regulation by HDL of the β2-Adrenergic Receptor Pathway. Implications in Selected Pathological Situations.Arch Med Res. 2015 Jul;46(5):361-71. doi: 10.1016/j.arcmed.2015.05.008. Epub 2015 May 23. Arch Med Res. 2015. PMID: 26009249 Review.
Cited by
-
Structural Insights into the Unexpected Agonism of Tetracyclic Antidepressants at Serotonin Receptors 5-HT1eR and 5-HT1FR.bioRxiv [Preprint]. 2023 Oct 7:2023.10.05.561100. doi: 10.1101/2023.10.05.561100. bioRxiv. 2023. Update in: Sci Adv. 2024 Apr 19;10(16):eadk4855. doi: 10.1126/sciadv.adk4855. PMID: 37986777 Free PMC article. Updated. Preprint.
-
Ligand-induced conformational changes in the β1-adrenergic receptor revealed by hydrogen-deuterium exchange mass spectrometry.Nat Commun. 2024 Oct 18;15(1):8993. doi: 10.1038/s41467-024-53161-0. Nat Commun. 2024. PMID: 39424782 Free PMC article.
-
A NanoBRET-Based H3R Conformational Biosensor to Study Real-Time H3 Receptor Pharmacology in Cell Membranes and Living Cells.Int J Mol Sci. 2022 Jul 26;23(15):8211. doi: 10.3390/ijms23158211. Int J Mol Sci. 2022. PMID: 35897787 Free PMC article.
-
Discovery of the cyclotide caripe 11 as a ligand of the cholecystokinin-2 receptor.Sci Rep. 2022 Jun 2;12(1):9215. doi: 10.1038/s41598-022-13142-z. Sci Rep. 2022. PMID: 35654807 Free PMC article.
-
Structural insights into the unexpected agonism of tetracyclic antidepressants at serotonin receptors 5-HT1eR and 5-HT1FR.Sci Adv. 2024 Apr 19;10(16):eadk4855. doi: 10.1126/sciadv.adk4855. Epub 2024 Apr 17. Sci Adv. 2024. PMID: 38630816 Free PMC article.
References
-
- Wootten D, Christopoulos A, Marti-Solano M, Babu MM, Sexton PM. Mechanisms of signalling and biased agonism in G protein-coupled receptors. Nat Rev Mol Cell Biol. 2018;19:638–653. - PubMed
-
- Shukla AK, Singh G, Ghosh E. Emerging structural insights into biased GPCR signaling. Trends Biochem Sci. 2014;39:594–602. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

