Membrane Proteomes and Ion Transporters in Bacillus anthracis and Bacillus subtilis Dormant and Germinating Spores
- PMID: 30602489
- PMCID: PMC6398275
- DOI: 10.1128/JB.00662-18
Membrane Proteomes and Ion Transporters in Bacillus anthracis and Bacillus subtilis Dormant and Germinating Spores
Abstract
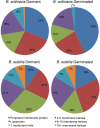
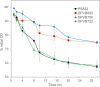
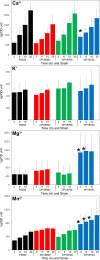
Bacterial endospores produced by Bacillus and Clostridium species can remain dormant and highly resistant to environmental insults for long periods, but they can also rapidly germinate in response to a nutrient-rich environment. Multiple proteins involved in sensing and responding to nutrient germinants, initiating solute and water transport, and accomplishing spore wall degradation are associated with the membrane surrounding the spore core. In order to more fully catalog proteins that may be involved in spore germination, as well as to identify protein changes taking place during germination, unbiased proteomic analyses of membrane preparations isolated from dormant and germinated spores of Bacillus anthracis and Bacillus subtilis were undertaken. Membrane-associated proteins were fractionated by SDS-PAGE, gel slices were trypsin digested, and extracted peptides were fractionated by liquid chromatography and analyzed by matrix-assisted laser desorption ionization-tandem time of flight mass spectrometry. More than 500 proteins were identified from each preparation. Bioinformatic methods were used to characterize proteins with regard to membrane association, cellular function, and conservation across species. Numerous proteins not previously known to be spore associated, 6 in B. subtilis and 68 in B. anthracis, were identified. Relative quantitation based on spectral counting indicated that the majority of spore membrane proteins decrease in abundance during the first 20 min of germination. The spore membranes contained several proteins thought to be involved in the transport of metal ions, a process that plays a major role in spore formation and germination. Analyses of mutant strains lacking these transport proteins implicated YloB in the accumulation of calcium within the developing forespore.IMPORTANCE Bacterial endospores can remain dormant and highly resistant to environmental insults for long periods but can also rapidly germinate in response to a nutrient-rich environment. The persistence and subsequent germination of spores contribute to their colonization of new environments and to the spread of certain diseases. Proteins of Bacillus subtilis and Bacillus anthracis were identified that are associated with the spore membrane, a position that can allow them to contribute to germination. A set of identified proteins that are predicted to carry out ion transport were examined for their contributions to spore formation, stability, and germination. Greater knowledge of spore formation and germination can contribute to the development of better decontamination strategies.
Keywords: Bacillus anthracis; germination; membrane; proteome; spore; subtilis.
Copyright © 2019 American Society for Microbiology.
Figures

Similar articles
-
Proteomic analysis of the spore coats of Bacillus subtilis and Bacillus anthracis.J Bacteriol. 2003 Feb;185(4):1443-54. doi: 10.1128/JB.185.4.1443-1454.2003. J Bacteriol. 2003. PMID: 12562816 Free PMC article.
-
Levels of germination proteins in Bacillus subtilis dormant, superdormant, and germinating spores.PLoS One. 2014 Apr 21;9(4):e95781. doi: 10.1371/journal.pone.0095781. eCollection 2014. PLoS One. 2014. PMID: 24752279 Free PMC article.
-
Identification of Bacillus anthracis proteins associated with germination and early outgrowth by proteomic profiling of anthrax spores.Proteomics. 2004 Sep;4(9):2653-61. doi: 10.1002/pmic.200400831. Proteomics. 2004. PMID: 15352240
-
Clostridium difficile spore biology: sporulation, germination, and spore structural proteins.Trends Microbiol. 2014 Jul;22(7):406-16. doi: 10.1016/j.tim.2014.04.003. Epub 2014 May 7. Trends Microbiol. 2014. PMID: 24814671 Free PMC article. Review.
-
Spore Germination.Microbiol Spectr. 2015 Dec;3(6). doi: 10.1128/microbiolspec.TBS-0014-2012. Microbiol Spectr. 2015. PMID: 27337279 Review.
Cited by
-
Gene Expression Analyses of Mutant Flammulina velutipes (Enokitake Mushroom) with Clogging Phenomenon.Mycobiology. 2022 Sep 19;50(5):366-373. doi: 10.1080/12298093.2022.2121497. eCollection 2022. Mycobiology. 2022. PMID: 36404905 Free PMC article.
-
The ionic liquid-assisted sample preparation method pTRUST allows sensitive proteome characterization of a variety of bacterial endospores to aid in the search for protein biomarkers.PLoS One. 2025 Jan 24;20(1):e0318186. doi: 10.1371/journal.pone.0318186. eCollection 2025. PLoS One. 2025. PMID: 39854521 Free PMC article.
-
The Membrane Proteome of Spores and Vegetative Cells of the Food-Borne Pathogen Bacillus cereus.Int J Mol Sci. 2021 Nov 19;22(22):12475. doi: 10.3390/ijms222212475. Int J Mol Sci. 2021. PMID: 34830357 Free PMC article.
-
A Family of Spore Lipoproteins Stabilizes the Germination Apparatus by Altering Inner Spore Membrane Fluidity in Bacillus subtilis Spores.J Bacteriol. 2023 Oct 26;205(10):e0014223. doi: 10.1128/jb.00142-23. Epub 2023 Jun 20. J Bacteriol. 2023. PMID: 37338384 Free PMC article.
-
Activation of the integrative and conjugative element Tn916 causes growth arrest and death of host bacteria.PLoS Genet. 2022 Oct 24;18(10):e1010467. doi: 10.1371/journal.pgen.1010467. eCollection 2022 Oct. PLoS Genet. 2022. PMID: 36279314 Free PMC article.
References
-
- Setlow P, Johnson EA. 2007. Spores and their significance, p 35–67. In Doyle MP, Beuchat LR (ed), Food microbiology: fundamentals and frontiers, 3rd ed ASM Press, Washington, DC.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

