Electrospun Collagen Nanofibers and Their Applications in Skin Tissue Engineering
- PMID: 30603521
- PMCID: PMC6171670
- DOI: 10.1007/s13770-017-0075-9
Electrospun Collagen Nanofibers and Their Applications in Skin Tissue Engineering
Abstract
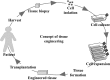
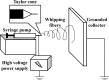
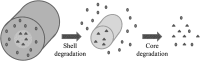
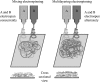
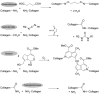
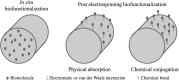
Electrospinning is a simple and versatile technique to fabricate continuous fibers with diameter ranging from micrometers to a few nanometers. To date, the number of polymers that have been electrospun has exceeded 200. In recent years, electrospinning has become one of the most popular scaffold fabrication techniques to prepare nanofiber mesh for tissue engineering applications. Collagen, the most abundant extracellular matrix protein in the human body, has been electrospun to fabricate biomimetic scaffolds that imitate the architecture of native human tissues. As collagen nanofibers are mechanically weak in nature, it is commonly cross-linked or blended with synthetic polymers to improve the mechanical strength without compromising the biological activity. Electrospun collagen nanofiber mesh has high surface area to volume ratio, tunable diameter and porosity, and excellent biological activity to regulate cell function and tissue formation. Due to these advantages, collagen nanofibers have been tested for the regeneration of a myriad of tissues and organs. In this review, we gave an overview of electrospinning, encompassing the history, the instrument settings, the spinning process and the parameters that affect fiber formation, with emphasis given to collagen nanofibers' fabrication and application, especially the use of collagen nanofibers in skin tissue engineering.
Keywords: Collagen; Electrospinning; Nanofiber; Scaffold; Skin; Tissue engineering.
Conflict of interest statement
The authors have no financial conflicts of interest.There is no animal experimental carried out for this article.
Figures


Similar articles
-
Engineering hybrid polymer-protein super-aligned nanofibers via rotary jet spinning.Biomaterials. 2014 Mar;35(10):3188-97. doi: 10.1016/j.biomaterials.2013.12.072. Epub 2014 Jan 20. Biomaterials. 2014. PMID: 24456606
-
Biomimetic electrospun nanofibers for tissue regeneration.Biomed Mater. 2006 Sep;1(3):R45-53. doi: 10.1088/1748-6041/1/3/R01. Epub 2006 Jul 28. Biomed Mater. 2006. PMID: 18458387 Review.
-
Electrospun Nanofiber Scaffolds and Their Hydrogel Composites for the Engineering and Regeneration of Soft Tissues.Methods Mol Biol. 2017;1570:261-278. doi: 10.1007/978-1-4939-6840-4_18. Methods Mol Biol. 2017. PMID: 28238143
-
The Use of Electrospinning Technique on Osteochondral Tissue Engineering.Adv Exp Med Biol. 2018;1058:247-263. doi: 10.1007/978-3-319-76711-6_11. Adv Exp Med Biol. 2018. PMID: 29691825 Review.
-
Advancements in electrospinning of polymeric nanofibrous scaffolds for tissue engineering.Tissue Eng Part B Rev. 2014 Aug;20(4):277-93. doi: 10.1089/ten.TEB.2013.0276. Epub 2013 Oct 12. Tissue Eng Part B Rev. 2014. PMID: 24004443 Review.
Cited by
-
Fabrication and Properties of Electrospun Collagen Tubular Scaffold Crosslinked by Physical and Chemical Treatments.Polymers (Basel). 2021 Feb 28;13(5):755. doi: 10.3390/polym13050755. Polymers (Basel). 2021. PMID: 33670963 Free PMC article.
-
A Review on Properties of Natural and Synthetic Based Electrospun Fibrous Materials for Bone Tissue Engineering.Membranes (Basel). 2018 Aug 14;8(3):62. doi: 10.3390/membranes8030062. Membranes (Basel). 2018. PMID: 30110968 Free PMC article. Review.
-
Recapitulating Cardiac Structure and Function In Vitro from Simple to Complex Engineering.Micromachines (Basel). 2021 Apr 1;12(4):386. doi: 10.3390/mi12040386. Micromachines (Basel). 2021. PMID: 33916254 Free PMC article. Review.
-
Tissue engineering applications of recombinant human collagen: a review of recent progress.Front Bioeng Biotechnol. 2024 Feb 14;12:1358246. doi: 10.3389/fbioe.2024.1358246. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38419725 Free PMC article. Review.
-
The Use of Natural Collagen Obtained from Fish Waste in Hair Styling and Care.Polymers (Basel). 2022 Feb 15;14(4):749. doi: 10.3390/polym14040749. Polymers (Basel). 2022. PMID: 35215659 Free PMC article.
References
-
- O’brien FJ. Biomaterials & scaffolds for tissue engineering. Mater Today. 2011;14:88–95. doi: 10.1016/S1369-7021(11)70058-X. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
