Full Thickness Skin Expansion ex vivo in a Newly Developed Reactor and Evaluation of Auto-Grafting Efficiency of the Expanded Skin Using Yucatan Pig Model
- PMID: 30603584
- PMCID: PMC6171704
- DOI: 10.1007/s13770-018-0154-6
Full Thickness Skin Expansion ex vivo in a Newly Developed Reactor and Evaluation of Auto-Grafting Efficiency of the Expanded Skin Using Yucatan Pig Model
Abstract
Background: Skin grafts are required in numerous clinical procedures, such as reconstruction after skin removal and correction of contracture or scarring after severe skin loss caused by burns, accidents, and trauma. The current standard for skin defect replacement procedures is the use of autologous skin grafts. However, donor-site tissue availability remains a major obstacle for the successful replacement of skin defects and often limits this option. The aim of this study is to effectively expand full thickness skin to clinically useful size using an automated skin reactor and evaluate auto grafting efficiency of the expanded skin using Yucatan female pigs.
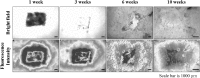
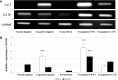
Methods: We developed an automated bioreactor system with the functions of real-time monitoring and remote-control, optimization of grip, and induction of skin porosity for effective tissue expansion. We evaluated the morphological, ultra-structural, and mechanical properties of the expanded skin before and after expansion using histology, immunohistochemistry, and tensile testing. We further carried out in vivo grafting study using Yucatan pigs to investigate the feasibility of this method in clinical application.
Results: The results showed an average expansion rate of 180%. The histological findings indicated that external expansion stimulated cellular activity in the isolated skin and resulted in successful grafting to the transplanted site. Specifically, hyperplasia did not appear at the auto-grafted site, and grafted skin appeared similar to normal skin. Furthermore, mechanical stimuli resulted in an increase in COL1A2 expression in a suitable environment.
Conclusions: These findings provided insight on the potential of this expansion system in promoting dermal extracellular matrix synthesis in vitro. Conclusively, this newly developed smart skin bioreactor enabled effective skin expansion ex vivo and successful grafting in vivo in a pig model.
Keywords: Animal model; Skin bioreactor; Skin transplantation; Tissue expansion devices.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.Animal care and experimental procedures were approved by the Institutional Animal Care and Use Committee at DGMIF (DGMIF-16100301-01, Daegu-Gyeongbuk Medical Innovation Foundation, Daegu, Korea).
Figures


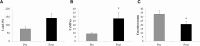





References
LinkOut - more resources
Full Text Sources
Miscellaneous
