Genetic labeling reveals temporal and spatial expression pattern of D2 dopamine receptor in rat forebrain
- PMID: 30604007
- PMCID: PMC6499762
- DOI: 10.1007/s00429-018-01824-2
Genetic labeling reveals temporal and spatial expression pattern of D2 dopamine receptor in rat forebrain
Abstract
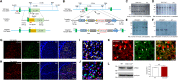
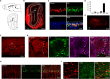
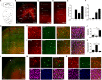
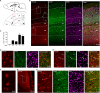
The D2 dopamine receptor (Drd2) is implicated in several brain disorders such as schizophrenia, Parkinson's disease, and drug addiction. Drd2 is also the primary target of both antipsychotics and Parkinson's disease medications. Although the expression pattern of Drd2 is relatively well known in mouse brain, the temporal and spatial distribution of Drd2 is lesser clear in rat brain due to the lack of Drd2 reporter rat lines. Here, we used CRISPR/Cas9 techniques to generate two knockin rat lines: Drd2::Cre and Rosa26::loxp-stop-loxp-tdTomato. By crossing these two lines, we produced Drd2 reporter rats expressing the fluorescence protein tdTomato under the control of the endogenous Drd2 promoter. Using fluorescence imaging and unbiased stereology, we revealed the cellular expression pattern of Drd2 in adult and postnatal rat forebrain. Strikingly, the Drd2 expression pattern differs between Drd2 reporter rats and Drd2 reporter mice generated by BAC transgene in prefrontal cortex and hippocampus. These results provide fundamental information needed for the study of Drd2 function in rat forebrain. The Drd2::Cre rats generated here may represent a useful tool to study the function of neuronal populations expressing Drd2.
Keywords: Cerebral cortex; Drd2; Hippocampus; Knockin rats; Olfactory bulb.
Conflict of interest statement
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee of East China Normal University.
Figures




Similar articles
-
CRISPR/Cas9-mediated targeting of the Rosa26 locus produces Cre reporter rat strains for monitoring Cre-loxP-mediated lineage tracing.FEBS J. 2017 Oct;284(19):3262-3277. doi: 10.1111/febs.14188. Epub 2017 Sep 19. FEBS J. 2017. PMID: 28763160
-
drd2-cre:ribotag mouse line unravels the possible diversity of dopamine d2 receptor-expressing cells of the dorsal mouse hippocampus.Hippocampus. 2015 Jul;25(7):858-75. doi: 10.1002/hipo.22408. Epub 2015 Feb 11. Hippocampus. 2015. PMID: 25545461
-
Striatal Distribution and Cytoarchitecture of Dopamine Receptor Subtype 1 and 2: Evidence from Double-Labeling Transgenic Mice.Front Neural Circuits. 2017 Aug 17;11:57. doi: 10.3389/fncir.2017.00057. eCollection 2017. Front Neural Circuits. 2017. PMID: 28860974 Free PMC article.
-
Inhibitory neuron-specific Cre-dependent red fluorescent labeling using VGAT BAC-based transgenic mouse lines with identified transgene integration sites.J Comp Neurol. 2018 Feb 15;526(3):373-396. doi: 10.1002/cne.24343. Epub 2017 Nov 11. J Comp Neurol. 2018. PMID: 29063602
-
A fresh look at cortical dopamine D2 receptor expressing neurons.Pharmacol Res. 2019 Jan;139:440-445. doi: 10.1016/j.phrs.2018.12.001. Epub 2018 Dec 4. Pharmacol Res. 2019. PMID: 30528973 Review.
Cited by
-
Advances in Genome Editing and Application to the Generation of Genetically Modified Rat Models.Front Genet. 2021 Apr 20;12:615491. doi: 10.3389/fgene.2021.615491. eCollection 2021. Front Genet. 2021. PMID: 33959146 Free PMC article. Review.
-
Retrograde Labeling Illuminates Distinct Topographical Organization of D1 and D2 Receptor-Positive Pyramidal Neurons in the Prefrontal Cortex of Mice.eNeuro. 2020 Oct 26;7(5):ENEURO.0194-20.2020. doi: 10.1523/ENEURO.0194-20.2020. Print 2020 Sep/Oct. eNeuro. 2020. PMID: 33037031 Free PMC article.
-
Bidirectional Regulation of Hippocampal Synaptic Plasticity and Modulation of Cumulative Spatial Memory by Dopamine D2-Like Receptors.Front Behav Neurosci. 2022 Jan 12;15:803574. doi: 10.3389/fnbeh.2021.803574. eCollection 2021. Front Behav Neurosci. 2022. PMID: 35095441 Free PMC article.
-
GRIN lens applications for studying neurobiology of substance use disorder.Addict Neurosci. 2022 Dec;4:100049. doi: 10.1016/j.addicn.2022.100049. Epub 2022 Nov 24. Addict Neurosci. 2022. PMID: 36531187 Free PMC article.
-
The Development of the Mesoprefrontal Dopaminergic System in Health and Disease.Front Neural Circuits. 2021 Oct 12;15:746582. doi: 10.3389/fncir.2021.746582. eCollection 2021. Front Neural Circuits. 2021. PMID: 34712123 Free PMC article. Review.
References
-
- Bean JC, Lin TW, Sathyamurthy A, Liu F, Yin DM, Xiong WC, Mei L. Genetic labeling reveals novel cellular targets of schizophrenia susceptibility gene: distribution of GABA and non-GABA ErbB4-positive cells in adult mouse brain. J Neurosci. 2014;34:13549–13566. doi: 10.1523/JNEUROSCI.2021-14.2014. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

