Site-specific effects of aromatase inhibition on the activation of male sexual behavior in male Japanese quail (Coturnix japonica)
- PMID: 30605622
- PMCID: PMC6377315
- DOI: 10.1016/j.yhbeh.2018.12.015
Site-specific effects of aromatase inhibition on the activation of male sexual behavior in male Japanese quail (Coturnix japonica)
Abstract
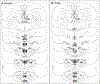
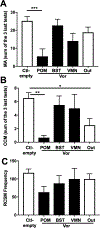
Aromatization within the medial preoptic nucleus (POM) is essential for the expression of male copulatory behavior in Japanese quail. However, several nuclei within the social behavior network (SBN) also express aromatase. Whether aromatase in these loci participates in the behavioral activation is not known. Castrated male Japanese quail were implanted with 2 subcutaneous Silastic capsules filled with crystalline testosterone and with bilateral stereotaxic implants filled with the aromatase inhibitor Vorozole targeting the POM, the bed nucleus of the stria terminalis (BST) or the ventromedial nucleus of the hypothalamus (VMN). Control animals were implanted with testosterone and empty bilateral stereotaxic implants. Starting 2 days after the surgery, subjects were tested for the expression of consummatory sexual behavior (CSB) every other day for a total of 10 tests. They were also tested once for appetitive sexual behavior (ASB) as measured by the rhythmic cloacal sphincter movements displayed in response to the visual presentation of a female. CSB was drastically reduced when the Vorozole implants were localized in the POM, but not in the BST nor in the VMN. Birds with implants in the BST took longer to show CSB in the first 6 tests than controls, suggesting a role of the BST in the acquisition of the full copulatory ability. ASB was not significantly affected by aromatase blockade in any region. These data confirm the key role played by the POM in the control of male sexual behavior and suggest a minor role for aromatization in the BST or VMN.
Keywords: Bed nucleus of the stria terminalis; Bird; Consummatory sexual behavior; Medial preoptic area; Neuroestrogens.
Copyright © 2019 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Effects of brain testosterone implants on appetitive and consummatory components of male sexual behavior in Japanese quail.Brain Res Bull. 1998 Sep 1;47(1):69-79. doi: 10.1016/s0361-9230(98)00064-1. Brain Res Bull. 1998. PMID: 9766392
-
Rapid effects of aromatase inhibition on male reproductive behaviors in Japanese quail.Horm Behav. 2006 Jan;49(1):45-67. doi: 10.1016/j.yhbeh.2005.05.003. Epub 2005 Jun 16. Horm Behav. 2006. PMID: 15963995 Free PMC article.
-
Role of aromatase in distinct brain nuclei of the social behaviour network in the expression of sexual behaviour in male Japanese quail.J Neuroendocrinol. 2022 Jun;34(6):e13127. doi: 10.1111/jne.13127. Epub 2022 Apr 8. J Neuroendocrinol. 2022. PMID: 35394094 Free PMC article.
-
Effects of testosterone and its metabolites on aromatase-immunoreactive cells in the quail brain: relationship with the activation of male reproductive behavior.J Steroid Biochem Mol Biol. 1996 Jan;56(1-6 Spec No):185-200. doi: 10.1016/0960-0760(95)00236-7. J Steroid Biochem Mol Biol. 1996. PMID: 8603040 Review.
-
The sexually dimorphic medial preoptic nucleus of quail: a key brain area mediating steroid action on male sexual behavior.Front Neuroendocrinol. 1996 Jan;17(1):51-125. doi: 10.1006/frne.1996.0002. Front Neuroendocrinol. 1996. PMID: 8788569 Review.
Cited by
-
Auditory learning in an operant task with social reinforcement is dependent on neuroestrogen synthesis in the male songbird auditory cortex.Horm Behav. 2020 May;121:104713. doi: 10.1016/j.yhbeh.2020.104713. Epub 2020 Feb 19. Horm Behav. 2020. PMID: 32057821 Free PMC article.
-
Brain Distribution and Sexually Dimorphic Expression of Amylin in Different Reproductive Stages of the Zebra Finch (Taeniopygia guttata) Suggest Roles of the Neuropeptide in Song Learning and Social Behaviour.Front Neurosci. 2020 Jan 13;13:1401. doi: 10.3389/fnins.2019.01401. eCollection 2019. Front Neurosci. 2020. PMID: 32009882 Free PMC article.
-
Exploring neuronal markers and early social environment influence in divergent quail lines selected for social motivation.Sci Rep. 2024 Oct 9;14(1):23554. doi: 10.1038/s41598-024-74906-3. Sci Rep. 2024. PMID: 39384852 Free PMC article.
-
A neural circuit perspective on brain aromatase.Front Neuroendocrinol. 2022 Apr;65:100973. doi: 10.1016/j.yfrne.2021.100973. Epub 2021 Dec 21. Front Neuroendocrinol. 2022. PMID: 34942232 Free PMC article. Review.
-
Effect of chronic intracerebroventricular administration of an aromatase inhibitor on the expression of socio-sexual behaviors in male Japanese quail.Behav Brain Res. 2021 Jul 23;410:113315. doi: 10.1016/j.bbr.2021.113315. Epub 2021 Apr 24. Behav Brain Res. 2021. PMID: 33901434 Free PMC article.
References
-
- Adkins-Regan EK, 2012. Hormonal organization and activation: Evolutionary implications and questions. Gen. Comp. Endocrinol 176, 279–285. - PubMed
-
- Adkins EK, 1977. Effects of diverse androgens on the sexual behavior and morphology of castrated male quail. Horm. Behav 8, 201–7. - PubMed
-
- Adkins EK, Adler NT, 1972. Hormonal control of behavior in the Japanese quail. J. Comp. Physiol. Psychol 81, 27–36. - PubMed
-
- Adkins EK, Boop JJ, Koutnik DL, Morris JB, Pniewski EE, 1980. Further evidence that androgen aromatization is essential for the activation of copulation in male quail. Physiol. Behav 24, 441–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

