Prevalence of BRCA1 and BRCA2 pathogenic and likely pathogenic variants in non-selected ovarian carcinoma patients in Brazil
- PMID: 30606148
- PMCID: PMC6319008
- DOI: 10.1186/s12885-018-5235-3
Prevalence of BRCA1 and BRCA2 pathogenic and likely pathogenic variants in non-selected ovarian carcinoma patients in Brazil
Abstract
Background: BRCA1/2 pathogenic (P) and likely pathogenic (LP) germline variants are frequent among patients with ovarian carcinoma. However, these variants have not been extensively characterized in patients with ovarian cancer in Brazil.
Methods: In this retrospective study we evaluated clinical characteristics and BRCA1/2 genetic test results from patients with ovarian carcinoma who underwent genetic counseling at A.C.Camargo Cancer Center (Brazil) between 2015 and 2017 and had performed germline genetic testing of BRCA1/2 genes.
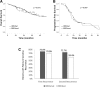
Results: Among 158 patients, 33 P and LP variants and were found (20.8%), 27 in BRCA1 and six in BRCA2, and six variants of unknown clinical significance (VUS). Thirteen percent of the patients did not have Multiplex Ligation-dependent Probe Amplification (MLPA) results. Three P variants in BRCA1 were found in more than one patient: c.5266dupC (p.Gln1756Profs*74), c.3331_3334delCAAG (p.Gln1111Asnfs5*), and c.211A > G (p.Arg71Gly). One LP variant in BRCA1 had not been previously described, c.4153_4154delCT (p.Leu1385Ilefs*5). Patients with previous diagnosis of breast cancer were carriers of P or LP variant in 8 of 12 cases (66.7%), and patients with a family history of ovarian or breast cancer in first- or second-degree relatives were carriers of P or LP variant in 26.7% of cases compared to 16.9% for patients without family history (p = 0.166).
Conclusion: Prevalence of BRCA1/2 germline P and LP variants is slightly higher than previously described by the largest occidental studies, with a high prevalence of variant c.5266dupC (p.Gln1756Profs*74) in BRCA1 observed. Moreover, we identified a new LP variant.
Keywords: BRCA1 and BRCA2 germline mutations; Brazil; Ovarian cancer; Prevalence.
Conflict of interest statement
Ethics approval and consent to participate
The A.C. Camargo Cancer Center Ethics Committee approved the study (CEP# 2450/17). The need for informed consent has been waived by the A.C. Camargo Cancer Center Ethics Committee.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


References
-
- Instituto Nacional De Câncer (Brasil) Estimativa 2018. Incidência do Câncer no Brasil. Rio de Janeiro: INCA; 2018.
-
- Rosenthal AN, Fraser LSM, Philpott S, Manchanda R, Burnell M, Badman P, et al. Evidence of stage shift in women diagnosed with ovarian Cancer during phase II of the United Kingdom familial ovarian Cancer screening study. J Clin Oncol. 2017;35:1411–1420. doi: 10.1200/JCO.2016.69.9330. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

