Astaxanthin Attenuates Environmental Tobacco Smoke-Induced Cognitive Deficits: A Critical Role of p38 MAPK
- PMID: 30609815
- PMCID: PMC6356379
- DOI: 10.3390/md17010024
Astaxanthin Attenuates Environmental Tobacco Smoke-Induced Cognitive Deficits: A Critical Role of p38 MAPK
Abstract
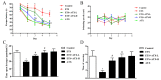
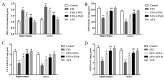
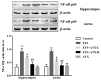
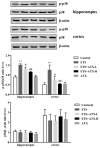
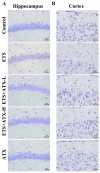
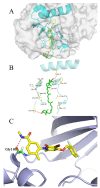
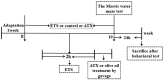
Increasing evidence indicates that environmental tobacco smoke (ETS) impairs cognitive function and induces oxidative stress in the brain. Recently, astaxanthin (ATX), a marine bioactive compound, has been reported to ameliorate cognitive deficits. However, the underlying pathogenesis remains unclear. In this study, ATX administration (40 mg/kg and 80 mg/kg, oral gavage) and cigarette smoking were carried out once a day for 10 weeks to investigate whether the p38 MAPK is involved in cognitive function in response to ATX treatment in the cortex and hippocampus of ETS mice. Results indicated that ATX administration improved spatial learning and memory of ETS mice (p < 0.05 or p < 0.01). Furthermore, exposure to ATX prevented the increases in the protein levels of the p38mitogen-activated protein kinase (p38 MAPK; p < 0.05 or p < 0.01) and nuclear factor-kappa B (NF-κB p65; p < 0.05 or p < 0.01), reversed the decreases in the mRNA and protein levels of synapsin I (SYN) and postsynaptic density protein 95 (PSD-95) (all p < 0.05 or p < 0.01). Moreover, ATX significantly down-regulated the increased levels of pro-inflammatory cytokines including interleukin-6 (IL-6) and tumor necrosis factor (TNF-α) (all p < 0.05 or p < 0.01). Meanwhile, the increased level of malondialdehyde (MDA) and the decreased activities of superoxide dismutase (SOD), glutathione (GSH), and catalase (CAT) were suppressed after exposure to ATX (all p < 0.05 or p < 0.01). Also, the results of the molecular docking study of ATX into the p38 MAPK binding site revealed that its mechanism was possibly similar to that of PH797804, a p38 MAPK inhibitor. Therefore, our results indicated that the ATX might be a critical agent in protecting the brain against neuroinflammation, synaptic plasticity impairment, and oxidative stress in the cortex and hippocampus of ETS mice.
Keywords: antioxidant inflammatory; astaxanthin; cigarette smoke exposure; p38 MAPK; synaptic-associated plasticity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
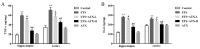


Similar articles
-
Aqueous extracts of se-enriched Auricularia auricular attenuates D-galactose-induced cognitive deficits, oxidative stress and neuroinflammation via suppressing RAGE/MAPK/NF-κB pathway.Neurosci Lett. 2019 Jun 21;704:106-111. doi: 10.1016/j.neulet.2019.04.002. Epub 2019 Apr 3. Neurosci Lett. 2019. PMID: 30953738
-
Grape Seed Proanthocyanidin Extract Ameliorates Streptozotocin-induced Cognitive and Synaptic Plasticity Deficits by Inhibiting Oxidative Stress and Preserving AKT and ERK Activities.Curr Med Sci. 2020 Jun;40(3):434-443. doi: 10.1007/s11596-020-2197-x. Epub 2020 Jul 17. Curr Med Sci. 2020. PMID: 32681248
-
[miR-155-5p alleviates lipopolysaccharide-induced inflammatory damage of human SH-SY5Y neuroblastoma cells by down-regulating SOCS1].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2023 Mar;39(3):220-229. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2023. PMID: 36946346 Chinese.
-
Oxidative stress-induced cognitive impairment in obesity can be reversed by vitamin D administration in rats.Nutr Neurosci. 2018 Dec;21(10):744-752. doi: 10.1080/1028415X.2017.1348436. Epub 2017 Jul 6. Nutr Neurosci. 2018. PMID: 28683595
-
Inhibition of p38 MAPK/NF-κB p65 signaling pathway activity by rare ginsenosides ameliorates cyclophosphamide-induced premature ovarian failure and KGN cell injury.J Ethnopharmacol. 2024 May 23;326:117944. doi: 10.1016/j.jep.2024.117944. Epub 2024 Feb 20. J Ethnopharmacol. 2024. PMID: 38382656
Cited by
-
Recent Advancements and Future Perspectives of Microalgae-Derived Pharmaceuticals.Mar Drugs. 2021 Dec 12;19(12):703. doi: 10.3390/md19120703. Mar Drugs. 2021. PMID: 34940702 Free PMC article. Review.
-
Tobacco and Nervous System Development and Function-New Findings 2015-2020.Brain Sci. 2021 Jun 16;11(6):797. doi: 10.3390/brainsci11060797. Brain Sci. 2021. PMID: 34208753 Free PMC article. Review.
-
Astaxanthin and its Effects in Inflammatory Responses and Inflammation-Associated Diseases: Recent Advances and Future Directions.Molecules. 2020 Nov 16;25(22):5342. doi: 10.3390/molecules25225342. Molecules. 2020. PMID: 33207669 Free PMC article. Review.
-
Biological activities of astaxanthin in the treatment of neurodegenerative diseases.Neurodegener Dis Manag. 2024;14(6):241-256. doi: 10.1080/17582024.2024.2433932. Epub 2024 Dec 8. Neurodegener Dis Manag. 2024. PMID: 39648516 Review.
-
Astaxanthin protects against environmentally persistent free radical-induced oxidative stress in well-differentiated respiratory epithelium.Redox Biol. 2025 Apr;81:103542. doi: 10.1016/j.redox.2025.103542. Epub 2025 Feb 9. Redox Biol. 2025. PMID: 39952200 Free PMC article.
References
MeSH terms
Substances
Grants and funding
- 81671069/National Natural Science Foundation of China
- JSKIM201703/Open Research Project of Jiangsu Key Laboratory of Immunity and Metabolism
- KC16SW164/Technology Innovation Project for the Special Social Development of Public Health in Xuzhou City
- KF201503/Open Program of Key Laboratory of Nuclear Medicine, and Ministry of Health and Jiangsu Key Laboratory of Molecular Nuclear Medicine
LinkOut - more resources
Full Text Sources
Miscellaneous

