Myelination in Multiple Sclerosis Lesions Is Associated with Regulation of Bone Morphogenetic Protein 4 and Its Antagonist Noggin
- PMID: 30609838
- PMCID: PMC6337410
- DOI: 10.3390/ijms20010154
Myelination in Multiple Sclerosis Lesions Is Associated with Regulation of Bone Morphogenetic Protein 4 and Its Antagonist Noggin
Abstract
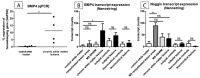
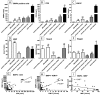
Remyelination is a central aspect of new multiple sclerosis (MS) therapies, in which one aims to alleviate disease symptoms by improving axonal protection. However, a central problem is mediators expressed in MS lesions that prevent effective remyelination. Bone morphogenetic protein4 (BMP4) inhibits the development of mature oligodendrocytes in cell culture and also blocks the expression of myelin proteins. Additionally, numerous studies have shown that Noggin (SYM1)-among other physiological antagonists of BMP4-plays a prominent role in myelin formation in the developing but also the adult central nervous system. Nonetheless, neither BMP4 nor Noggin have been systematically studied in human MS lesions. In this study, we demonstrated by transcript analysis and immunohistochemistry that BMP4 is expressed by astrocytes and microglia/macrophages in association with inflammatory infiltrates in MS lesions, and that astrocytes also express BMP4 in chronic inactive lesions that failed to remyelinate. Furthermore, the demonstration of an increased expression of Noggin in so-called shadow plaques (i.e., remyelinated lesions with thinner myelin sheaths) in comparison to chronically inactive demyelinated lesions implies that antagonizing BMP4 is associated with successful remyelination in MS plaques in humans. However, although BMP4 is strongly overexpressed in inflammatory lesion areas, its levels are also elevated in remyelinated lesion areas, which raises the possibility that BMP4 signaling itself may be required for remyelination. Therefore, remyelination might be influenced by a small number of key factors. Manipulating these molecules, i.e., BMP4 and Noggin, could be a promising therapeutic approach for effective remyelination.
Keywords: BMP4; Noggin; bone morphogenetic protein 4; multiple sclerosis; remyelination; remyelination block.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Bone Morphogenetic Proteins: Inhibitors of Myelination in Development and Disease.Vitam Horm. 2015;99:195-222. doi: 10.1016/bs.vh.2015.05.005. Vitam Horm. 2015. PMID: 26279377 Review.
-
Investigation of sequential growth factor delivery during cuprizone challenge in mice aimed to enhance oligodendrogliogenesis and myelin repair.PLoS One. 2013 May 1;8(5):e63415. doi: 10.1371/journal.pone.0063415. Print 2013. PLoS One. 2013. PMID: 23650566 Free PMC article.
-
Transcript profiling of different types of multiple sclerosis lesions yields FGF1 as a promoter of remyelination.Acta Neuropathol Commun. 2014 Dec 11;2:168. doi: 10.1186/s40478-014-0168-9. Acta Neuropathol Commun. 2014. PMID: 25589163 Free PMC article.
-
Inhibiting Bone Morphogenetic Protein 4 Type I Receptor Signaling Promotes Remyelination by Potentiating Oligodendrocyte Differentiation.eNeuro. 2019 May 14;6(2):ENEURO.0399-18.2019. doi: 10.1523/ENEURO.0399-18.2019. Print 2019 Mar/Apr. eNeuro. 2019. PMID: 31028086 Free PMC article.
-
Macroglial diversity: white and grey areas and relevance to remyelination.Cell Mol Life Sci. 2021 Jan;78(1):143-171. doi: 10.1007/s00018-020-03586-9. Epub 2020 Jul 9. Cell Mol Life Sci. 2021. PMID: 32648004 Free PMC article. Review.
Cited by
-
Inhibitors of Myelination and Remyelination, Bone Morphogenetic Proteins, are Upregulated in Human Neurological Disease.Neurochem Res. 2020 Mar;45(3):656-662. doi: 10.1007/s11064-020-02980-w. Epub 2020 Feb 6. Neurochem Res. 2020. PMID: 32030597 Free PMC article. Review.
-
CD8+ T cell gene expression analysis identifies differentially expressed genes between multiple sclerosis patients and healthy controls.Mult Scler J Exp Transl Clin. 2020 Dec 9;6(4):2055217320978511. doi: 10.1177/2055217320978511. eCollection 2020 Oct-Dec. Mult Scler J Exp Transl Clin. 2020. PMID: 33343920 Free PMC article.
-
Noggin alleviates neuropathic pain by regulating microglial polarization and remodeling iron homeostasis via a STAT3-dependent pathway.Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2025 Apr 28;50(4):602-614. doi: 10.11817/j.issn.1672-7347.2025.250206. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2025. PMID: 40785674 Free PMC article. Chinese, English.
-
Infiltrating circulating monocytes provide an important source of BMP4 at the early stage of spinal cord injury.Dis Model Mech. 2023 Jan 1;16(1):dmm049856. doi: 10.1242/dmm.049856. Epub 2023 Jan 18. Dis Model Mech. 2023. PMID: 36518009 Free PMC article.
-
Combinatory Multifactor Treatment Effects on Primary Nanofiber Oligodendrocyte Cultures.Cells. 2019 Nov 12;8(11):1422. doi: 10.3390/cells8111422. Cells. 2019. PMID: 31726669 Free PMC article.
References
-
- Hagemeier K., Bruck W., Kuhlmann T. Multiple sclerosis—Remyelination failure as a cause of disease progression. Histol. Histopathol. 2012;27:277–287. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

