Combinatorial pathway enzyme engineering and host engineering overcomes pyruvate overflow and enhances overproduction of N-acetylglucosamine in Bacillus subtilis
- PMID: 30609921
- PMCID: PMC6318901
- DOI: 10.1186/s12934-018-1049-x
Combinatorial pathway enzyme engineering and host engineering overcomes pyruvate overflow and enhances overproduction of N-acetylglucosamine in Bacillus subtilis
Abstract
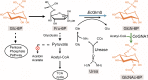
Background: Glucosamine-6-phosphate N-acetyltransferase (GNA1) is the key enzyme that causes overproduction of N-acetylglucosamine in Bacillus subtilis. Previously, we increased GlcNAc production by promoting the expression of GNA1 from Caenorhabditis elegans (CeGNA1) in an engineered B. subtilis strain BSGN12. In this strain overflow metabolism to by-products acetoin and acetate had been blocked by mutations, however pyruvate accumulated as an overflow metabolite. Although overexpression of CeGNA1 drove carbon flux from pyruvate to the GlcNAc synthesis pathway and decreased pyruvate accumulation, the residual pyruvate reduced the intracellular pH, resulting in inhibited CeGNA1 activity and limited GlcNAc production.
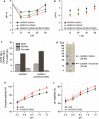
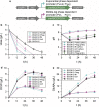
Results: In this study, we attempted to further overcome pyruvate overflow by enzyme engineering and host engineering for enhanced GlcNAc production. To this end, the key enzyme CeGNA1 was evolved through error-prone PCR under pyruvate stress to enhance its catalytic activity. Then, the urease from Bacillus paralicheniformis was expressed intracellularly to neutralize the intracellular pH, making it more robust in growth and more efficient in GlcNAc production. It was found that the activity of mutant CeGNA1 increased by 11.5% at pH 6.5-7.5, with the catalytic efficiency increasing by 27.5% to 1.25 s-1 µM-1. Modulated expression of urease increased the intracellular pH from 6.0 to 6.8. The final engineered strain BSGN13 overcame pyruvate overflow, produced 25.6 g/L GlcNAc with a yield of 0.43 g GlcNAc/g glucose in a shake flask fermentation and produced 82.5 g/L GlcNAc with a yield of 0.39 g GlcNAc/g glucose by fed-batch fermentation, which was 1.7- and 1.2-times, respectively, of the yield achieved previously.
Conclusions: This study highlights a strategy that combines pathway enzyme engineering and host engineering to resolve overflow metabolism in B. subtilis for the overproduction of GlcNAc. By means of modulated expression of urease reduced pyruvate burden, conferred bacterial survival fitness, and enhanced GlcNAc production, all of which improved our understanding of co-regulation of cell growth and metabolism to construct more efficient B. subtilis cell factories.
Keywords: Bacillus subtilis; Glucosamine-6-phosphate N-acetyltransferase; N-Acetylglucosamine; Overflow; Pyruvate; Urease.
Figures


Similar articles
-
Pathway engineering of Bacillus subtilis for microbial production of N-acetylglucosamine.Metab Eng. 2013 Sep;19:107-15. doi: 10.1016/j.ymben.2013.07.002. Epub 2013 Jul 20. Metab Eng. 2013. PMID: 23876412
-
Combinatorial Fine-Tuning of GNA1 and GlmS Expression by 5'-Terminus Fusion Engineering Leads to Overproduction of N-Acetylglucosamine in Bacillus subtilis.Biotechnol J. 2019 Mar;14(3):e1800264. doi: 10.1002/biot.201800264. Epub 2018 Sep 4. Biotechnol J. 2019. PMID: 30105781
-
Rewiring the Glucose Transportation and Central Metabolic Pathways for Overproduction of N-Acetylglucosamine in Bacillus subtilis.Biotechnol J. 2017 Oct;12(10). doi: 10.1002/biot.201700020. Epub 2017 Aug 14. Biotechnol J. 2017. PMID: 28731580
-
[Strategies and tools for metabolic engineering in Bacillus subtilis].Sheng Wu Gong Cheng Xue Bao. 2021 May 25;37(5):1619-1636. doi: 10.13345/j.cjb.200543. Sheng Wu Gong Cheng Xue Bao. 2021. PMID: 34085446 Review. Chinese.
-
Microbial production of N-acetyl-D-glucosamine (GlcNAc) for versatile applications: Biotechnological strategies for green process development.Carbohydr Res. 2024 Feb;536:109039. doi: 10.1016/j.carres.2024.109039. Epub 2024 Jan 17. Carbohydr Res. 2024. PMID: 38277719 Review.
Cited by
-
Design of a programmable biosensor-CRISPRi genetic circuits for dynamic and autonomous dual-control of metabolic flux in Bacillus subtilis.Nucleic Acids Res. 2020 Jan 24;48(2):996-1009. doi: 10.1093/nar/gkz1123. Nucleic Acids Res. 2020. PMID: 31799627 Free PMC article.
-
Comparative effects of alive and pasteurized Akkermansia muciniphila on normal diet-fed mice.Sci Rep. 2021 Sep 9;11(1):17898. doi: 10.1038/s41598-021-95738-5. Sci Rep. 2021. PMID: 34504116 Free PMC article.
-
Understanding the limitations of substrate degradation in bioelectrochemical systems.Front Microbiol. 2025 Jan 6;15:1511142. doi: 10.3389/fmicb.2024.1511142. eCollection 2024. Front Microbiol. 2025. PMID: 39834375 Free PMC article.
-
Gut bacteriome dynamics in high altitude-adapted chicken lines: a key to future poultry therapeutics.Sci Rep. 2025 Apr 7;15(1):11910. doi: 10.1038/s41598-025-96178-1. Sci Rep. 2025. PMID: 40195460 Free PMC article.
-
Vitreoscilla Haemoglobin: A Tool to Reduce Overflow Metabolism.Microorganisms. 2021 Dec 26;10(1):43. doi: 10.3390/microorganisms10010043. Microorganisms. 2021. PMID: 35056491 Free PMC article. Review.
References
-
- Liu YF, Zhu YQ, Ma WL, Shin HD, Li JH, Liu L, Du GC, Chen J. Spatial modulation of key pathway enzymes by DNA-guided scaffold system and respiration chain engineering for improved N-acetylglucosamine production by Bacillus subtilis. Metab Eng. 2014;24:61–69. doi: 10.1016/j.ymben.2014.04.004. - DOI - PubMed
-
- Dorfmueller HC, Fang W, Rao FV, Blair DE, Attrill H, van Aalten DM. Structural and biochemical characterization of a trapped coenzyme A adduct of Caenorhabditis elegans glucosamine-6-phosphate N-acetyltransferase 1. Acta Crystallogr D Biol Crystallogr. 2012;68:1019–1029. doi: 10.1107/S0907444912019592. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

