Safety and efficacy of infliximab in the treatment of refractory uveoretinitis in Behçet's disease: a large-scale, long-term postmarketing surveillance in Japan
- PMID: 30611312
- PMCID: PMC6321670
- DOI: 10.1186/s13075-018-1793-7
Safety and efficacy of infliximab in the treatment of refractory uveoretinitis in Behçet's disease: a large-scale, long-term postmarketing surveillance in Japan
Abstract
Background: Infliximab, an anti-tumor necrosis factor-alpha antibody, has been reported to have excellent efficacy for refractory uveoretinitis in Behçet's disease (RUBD), and was approved for this indication in Japan. However, the long-term safety profile and efficacy in real-world clinical settings in patients with RUBD have not been fully clarified. The BRIGHT study, a prospective, large-scale, long-term postmarketing surveillance (PMS) study, was conducted to investigate the long-term safety and efficacy of infliximab in Japanese patients with RUBD.
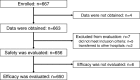
Methods: All patients with RUBD who started infliximab treatment between January 2007 and January 2010 were enrolled. Safety was evaluated every 6 months for up to 24 months after initiation of therapy in 656 patients, and efficacy was evaluated in 650 patients. Patient characteristics were compared using the chi-square or Fisher's exact test. The frequency of ocular attacks before and after infliximab treatment was compared using the Wilcoxon signed-rank test. Independent associated factors for safety or efficacy were identified using multiple logistic regression analysis. A two-sided p value <0.05 was considered significant.
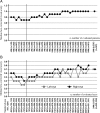
Results: Among the 656 patients evaluated for safety, 555 (84.6%) completed the 24-month study period. The incidence of adverse drug reactions (ADRs) and serious ADRs were 32.32% and 6.10%, respectively, and the safety profile was comparable to that of Japanese PMS of infliximab for other diseases. The most common ADRs and serious ADRs were infections (11.89% and 3.66%). Tuberculosis was reported in two patients, and Pneumocystis jirovecii in one. Identified independent associated factors for infections were comorbid respiratory disease, history of allergic disease, and concomitant use of glucocorticoids. Although infusion reactions were observed in 11.13% of patients, most were non-serious. The response rate at 24 months by physician global assessment was 80.7%. Median frequency of ocular attacks per 6 months significantly decreased compared with that before infliximab treatment (2.0 to 0.0), and corrected visual acuity was maintained during the study.
Conclusions: Infliximab treatment had good tolerability and efficacy in Japanese patients with RUBD in this large-scale, long-term PMS. Infliximab treatment seemed to be a good treatment option for RUBD in real-world clinical settings.
Trial registration: UMIN Clinical Trials Registry, UMIN000027733 . Retrospectively registered on 6 June 2017.
Keywords: Associated factor; Behçet’s disease; Biological therapies; Efficacy; Infliximab; Postmarketing surveillance; Safety; Uveoretinitis.
Conflict of interest statement
Ethics approval and consent to participate
This PMS study protocol was approved by the Ministry of Health, Labor and Welfare of the Japan Government, and was conducted in accordance with good post-marketing surveillance practice (GPSP), which sets the standard for PMS studies of approved drugs in clinical practice. Patients were treated in accordance with physicians’ usual clinical practice and indication for commercially available IFX.; no consent was needed by the institutions’ ethics committees.
Consent for publication
Not applicable.
Competing interests
SO has received expert testimony fees from Mitsubishi Tanabe Pharma Corporation, AbbVie GK, Alcon Japan, Bosch & Lomb Japan, and Santen Pharmaceutical Company; grants/research support from Mitsubishi Tanabe Pharma Corporation; lecture fees from Mitsubishi Tanabe Pharma Corporation, Alcon Japan, Otsuka Pharmaceutical Company, and Santen Pharmaceutical Company; patent fees from Mitsubishi Tanabe Pharma Corporation. IU, MM, TG, and TY are full-time employees of Mitsubishi Tanabe Pharma Corporation.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Efficacy, safety, and pharmacokinetics of multiple administration of infliximab in Behçet's disease with refractory uveoretinitis.J Rheumatol. 2004 Jul;31(7):1362-8. J Rheumatol. 2004. PMID: 15229958
-
[Effects and safety of infliximab administration in refractory uveoretinitis with Behçet's disease].Nippon Ganka Gakkai Zasshi. 2010 Feb;114(2):87-95. Nippon Ganka Gakkai Zasshi. 2010. PMID: 20187505 Japanese.
-
Real-World Safety and Effectiveness of Infliximab in 255 Patients with Intestinal, Neurological, and Vascular Behçet's Disease: A Post-Marketing Surveillance.Adv Ther. 2024 Dec;41(12):4476-4497. doi: 10.1007/s12325-024-02993-9. Epub 2024 Oct 16. Adv Ther. 2024. PMID: 39412631 Free PMC article.
-
Long-term efficacy, safety, and cumulative retention rate of antitumor necrosis factor-alpha treatment for patients with Behcet's uveitis: A systematic review and meta-analysis.Int J Rheum Dis. 2024 Feb;27(2):e15096. doi: 10.1111/1756-185X.15096. Int J Rheum Dis. 2024. PMID: 38402428
-
Evaluation of disease activity in uveoretinitis associated with Behçet's disease.Immunol Med. 2021 Jun;44(2):86-97. doi: 10.1080/25785826.2020.1800244. Epub 2020 Jul 29. Immunol Med. 2021. PMID: 32726184 Review.
Cited by
-
Topical Ocular Anti-TNFα Agent Licaminlimab in the Treatment of Acute Anterior Uveitis: A Randomized Phase II Pilot Study.Transl Vis Sci Technol. 2022 Jun 1;11(6):14. doi: 10.1167/tvst.11.6.14. Transl Vis Sci Technol. 2022. PMID: 35704329 Free PMC article. Clinical Trial.
-
A 10-year follow-up of infliximab monotherapy for refractory uveitis in Behçet's syndrome.Sci Rep. 2020 Dec 17;10(1):22227. doi: 10.1038/s41598-020-78718-z. Sci Rep. 2020. PMID: 33335139 Free PMC article.
-
Microbiome in Immune-Mediated Uveitis.Int J Mol Sci. 2022 Jun 24;23(13):7020. doi: 10.3390/ijms23137020. Int J Mol Sci. 2022. PMID: 35806031 Free PMC article. Review.
-
Perspectives of Therapeutic Drug Monitoring of Biological Agents in Non-Infectious Uveitis Treatment: A Review.Pharmaceutics. 2023 Feb 25;15(3):766. doi: 10.3390/pharmaceutics15030766. Pharmaceutics. 2023. PMID: 36986627 Free PMC article. Review.
-
Network meta-analysis of immunomodulatory therapies for Behçet's disease-associated uveitis: comparative evaluation of safety and efficacy profiles.Graefes Arch Clin Exp Ophthalmol. 2025 Aug 19. doi: 10.1007/s00417-025-06938-y. Online ahead of print. Graefes Arch Clin Exp Ophthalmol. 2025. PMID: 40828404
References
-
- Nakamura S, Yamakawa T, Sugita M, Kijima M, Ishioka M, Tanaka S, et al. The role of tumor necrosis factor-alpha in the induction of experimental autoimmune uveoretinitis in mice. Invest Ophthalmol Vis Sci. 1994;35:3884–3889. - PubMed
-
- Sartani G, Silver PB, Rizzo LV, Chan CC, Wiggert B, Mastorakos G, et al. Anti-tumor necrosis factor alpha therapy suppresses the induction of experimental autoimmune uveoretinitis in mice by inhibiting antigen priming. Invest Ophthalmol Vis Sci. 1996;37:2211–2218. - PubMed
-
- Nakamura S, Sugita M, Tanaka S, Ohno S. Enhanced production of in vitro tumor necrosis factor-alpha from monocytes in Behçet’s disease. Nippon Ganka Gakkai Zasshi. 1992;96:1282–1285. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Medical