Hydroxymethanesulfonate from Volcanic Sulfur Dioxide: A "Mineral" Reservoir for Formaldehyde and Other Simple Carbohydrates in Prebiotic Chemistry
- PMID: 30615473
- PMCID: PMC6459274
- DOI: 10.1089/ast.2017.1800
Hydroxymethanesulfonate from Volcanic Sulfur Dioxide: A "Mineral" Reservoir for Formaldehyde and Other Simple Carbohydrates in Prebiotic Chemistry
Abstract
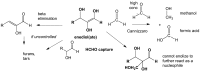
While formaldehyde (HCHO) was likely generated in Earth's prebiotic atmosphere by ultraviolet light, electrical discharge, and/or volcano-created lightning, HCHO could not have accumulated in substantial amounts in prebiotic environments, including those needed for prebiotic processes that generate nucleosidic carbohydrates. HCHO at high concentrations in alkaline solutions self-reacts in the Cannizzaro reaction to give methanol and formate, neither having prebiotic value. Here, we explore the possibility that volcanic sulfur dioxide (SO2) might have generated a reservoir for Hadean HCHO by a reversible reaction with HCHO to give hydroxymethanesulfonate (HMS). We show that salts of HMS are stable as solids at 90°C and do not react with themselves in solution, even at high (>8 M) concentrations. This makes them effective stores of HCHO, since the reverse reaction slowly delivers HCHO back into an environment where it can participate in prebiotically useful reactions. Specifically, we show that in alkaline borate solutions, HCHO derived from HMS allows formation of borate-stabilized carbohydrates as effectively as free HCHO, without losing material to Cannizzaro products. Further, we show that SO2 can perform similar roles for glycolaldehyde and glyceraldehyde, two intrinsically unstable carbohydrates that are needed by various models as precursors for RNA building blocks. Zircons from the Hadean show that the Hadean mantle likely provided volcanic SO2 at rates at least as great as the rates of atmospheric HCHO generation, making the formation of Hadean HMS essentially unavoidable. Thus, hydroxymethylsulfonate adducts of formaldehyde, glycolaldehyde, and glyceraldehyde, including the less soluble barium, strontium, and calcium salts, are likely candidates for prebiotically useful organic minerals on early Earth.
Keywords: Borate; Cannizzaro reaction; Formaldehyde; Hydroxymethanesulfonate; Organic minerals; Origins of life; Prebiotic chemistry.; Sulfur dioxide.
Figures





References
-
- Amaral A.F., Marques M.M., da Silva J.A.L., and da Silva J.J.R.F. (2008) Interactions of D-ribose with polyatomic anions, and alkaline and alkaline-earth cations: possible clues to environmental synthesis conditions in the pre-RNA world. New Journal of Chemistry 32:2043–2049
-
- Andres R.J. and Kasgnoc A.D. (1998) Time-Averaged Inventory of Volcanic Sulfur Emissions [online dataset]. Global Emissions Inventory Activity, Version 1. https://gcmd.nasa.gov/KeywordSearch/Metadata.do?Portal=idn_ceos&KeywordP...
-
- Appayee C. and Breslow R. (2014) Deuterium studies reveal a new mechanism for the formose reaction involving hydride shifts. J Am Chem Soc 136:3720–3723 - PubMed
-
- Becker S., Thoma I., Deutsch A., Gehrke T., Mayer P., Zipse H., and Carell T. (2016) A high-yielding, strictly regioselective prebiotic purine nucleoside formation pathway. Science 352:833–836 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
