Romantic Surfaces: A Systematic Overview of Stable, Biospecific, and Antifouling Zwitterionic Surfaces
- PMID: 30620199
- PMCID: PMC6365910
- DOI: 10.1021/acs.langmuir.8b03360
Romantic Surfaces: A Systematic Overview of Stable, Biospecific, and Antifouling Zwitterionic Surfaces
Abstract
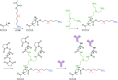
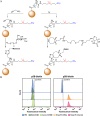
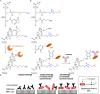
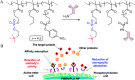
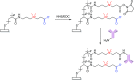
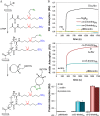
This Feature Article focuses on recent advances in the bioconjugation of surface-bound zwitterionic polymers for biospecific antifouling surfaces. Various approaches for the functionalization of antifouling zwitterionic polymers are systematically investigated, such as chain-end and side-chain functionalization. Side-chain functionalization methods can be further classified as those that are achieved through homopolymerization of custom-synthesized zwitterionic monomers equipped with reactive groups, or those that are achieved via synthesis of random or block copolymers combining different monomers with antifouling functionality and others with reactive groups. Several of the pros and cons of these approaches are outlined and discussed. Finally, some perspective and future directions of research are presented toward long-term stable, generically repelling surfaces that strongly and specifically adhere to a single component in a complex mixture.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




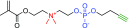
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

