Acute Myeloid Leukemia Affects Mouse Sperm Parameters, Spontaneous Acrosome Reaction, and Fertility Capacity
- PMID: 30626098
- PMCID: PMC6337746
- DOI: 10.3390/ijms20010219
Acute Myeloid Leukemia Affects Mouse Sperm Parameters, Spontaneous Acrosome Reaction, and Fertility Capacity
Abstract
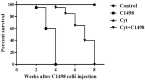
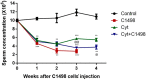
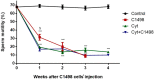
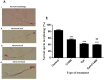
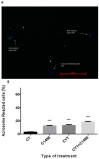
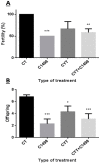
Leukemia is one of the most common cancers in patients of reproductive age. It is well known that chemotherapy, used as anti-cancer therapy, adversely affects male fertility. Moreover, the negative effect of leukemia on sperm quality, even before chemotherapy treatment, has been reported. However, the mechanisms behind this disease's effect on sperm quality remains unknown. In this study, we examine the direct effect of leukemia and chemotherapy alone and in combination on sperm parameters and male fertility. For this, we developed an acute myeloid leukemia (AML) mouse model (mice were treated with AML cells C1498 and developed leukemia); these mice then received cytarabine chemotherapy. Our findings reveal a significant reduction in sperm concentration and motility and a significant increase in abnormal morphology and spontaneous acrosome reaction of the sperm following AML and chemotherapy treatment, alone and in combination. We also found a reduction in male fertility and the number of delivered offspring. Our results support previous findings that AML impairs sperm parameters and show for the first time that AML increases spontaneous acrosome reaction and decreases male fertility capacity and number of offspring.
Keywords: acrosome reaction; acute myeloid leukemia; male infertility; sperm parameters; testis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Granulocyte Colony-Stimulating Factor Restored Impaired Spermatogenesis and Fertility in an AML-Chemotherapy Mice Model.Int J Mol Sci. 2021 Oct 15;22(20):11157. doi: 10.3390/ijms222011157. Int J Mol Sci. 2021. PMID: 34681817 Free PMC article.
-
Effects of sulfasalazine on sperm acrosome reaction and gene expression in the male reproductive organs of rats.Toxicol Sci. 2005 May;85(1):675-82. doi: 10.1093/toxsci/kfi071. Epub 2004 Dec 29. Toxicol Sci. 2005. PMID: 15625186
-
Human sperm Toll-like receptor 4 (TLR4) mediates acrosome reaction, oxidative stress markers, and sperm parameters in response to bacterial lipopolysaccharide in infertile men.J Assist Reprod Genet. 2017 Aug;34(8):1067-1077. doi: 10.1007/s10815-017-0957-8. Epub 2017 May 26. J Assist Reprod Genet. 2017. PMID: 28550386 Free PMC article.
-
Toxic effects of 2,4-dichlorophenoxyacetic acid on human sperm function in vitro.J Toxicol Sci. 2016;41(4):543-9. doi: 10.2131/jts.41.543. J Toxicol Sci. 2016. PMID: 27432240
-
Sperm function tests and fertility.Int J Androl. 2006 Feb;29(1):69-75; discussion 105-8. doi: 10.1111/j.1365-2605.2005.00630.x. Int J Androl. 2006. PMID: 16466526 Review.
Cited by
-
Maternal supplementation of alpha-lipoic acid ameliorates prenatal cytarabine-induced mutilation in reproductive development and function in F1 male adult rats.Naunyn Schmiedebergs Arch Pharmacol. 2024 Jun;397(6):4035-4053. doi: 10.1007/s00210-023-02852-4. Epub 2023 Nov 27. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 38010397
-
Effect of Chemotherapy Cytarabine and Acute Myeloid Leukemia on the Development of Spermatogenesis at the Adult Age of Immature Treated Mice.Int J Mol Sci. 2022 Apr 4;23(7):4013. doi: 10.3390/ijms23074013. Int J Mol Sci. 2022. PMID: 35409373 Free PMC article.
-
Granulocyte Colony-Stimulating Factor Restored Impaired Spermatogenesis and Fertility in an AML-Chemotherapy Mice Model.Int J Mol Sci. 2021 Oct 15;22(20):11157. doi: 10.3390/ijms222011157. Int J Mol Sci. 2021. PMID: 34681817 Free PMC article.
-
Approaches and Technologies in Male Fertility Preservation.Int J Mol Sci. 2020 Jul 31;21(15):5471. doi: 10.3390/ijms21155471. Int J Mol Sci. 2020. PMID: 32751826 Free PMC article. Review.
-
Advancements in leukemia management: Bridging diagnosis, prognosis and nanotechnology (Review).Int J Oncol. 2024 Dec;65(6):112. doi: 10.3892/ijo.2024.5700. Epub 2024 Oct 4. Int J Oncol. 2024. PMID: 39364739 Free PMC article. Review.
References
-
- Bramswig J.H., Heimes U., Heiermann E., Schlegel W., Nieschlag E., Schellong G. The effects of different cumulative doses of chemotherapy on testicular function. Results in 75 patients treated for Hodgkin’s disease during childhood or adolescence. Cancer. 1990;65:1298–1302. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

