Event-free survival following early endometrial events in breast cancer patients treated with anti-hormonal therapy: A nationwide claims data study
- PMID: 30633178
- PMCID: PMC6336644
- DOI: 10.1097/MD.0000000000013976
Event-free survival following early endometrial events in breast cancer patients treated with anti-hormonal therapy: A nationwide claims data study
Abstract
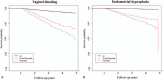
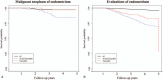
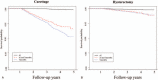
Tamoxifen, an anti-estrogen agent that can suppress breast cancer, has been reported to increase endometrium-related adverse events. There are no guidelines for screening tamoxifen-treated patients for endometrial disease. We analyzed nationwide claims data related to endometrial diseases to investigate patterns of endometrial disease in breast cancer patients who underwent hormonal treatment.We sourced claims data from the Health Insurance Review and Assessment Service in South Korea. Patients who made their first claim for an anti-hormonal agent between January 1, 2010 and December 31, 2012 were enrolled retrospectively. We analyzed patient characteristics and all claims related to endometrial disease, stratified by prescribed hormonal agents.Among a total of 32,496 enrolled patients, 19,603 used tamoxifen only and 10,101 were treated with an aromatase inhibitor (AI) alone. Endometrial events occurred in 15.4% (3028/19603) of the tamoxifen-only patients and 2.0% (201/10101) of the AI-only group. In patients diagnosed with breast cancer at the age of 50 or older, the hazard ratio (HR) of endometrial malignancy in the tamoxifen-only group compared to the AI-only group was 4.13 (95% CI 1.404-12.159, P = .010). The HR of curettage in the tamoxifen-only group was 31.0 (95% CI 19.668-48.831, P <.001).The occurrence of endometrial events among tamoxifen-treated breast cancer patients was higher than in patients treated with only AI, similar to previous studies. However, the HR of curettage was uniquely high, despite its invasiveness. Guidelines for screening endometrial disease and improvements of healthcare policy are required to appropriately manage high-risk patients.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures

References
-
- Berube S, Provencher L, Robert J, et al. Quantitative exploration of possible reasons for the recent improvement in breast cancer survival. Breast Cancer Res Treat 2007;106:419–31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

