Rab32 interacts with SNX6 and affects retromer-dependent Golgi trafficking
- PMID: 30640902
- PMCID: PMC6331118
- DOI: 10.1371/journal.pone.0208889
Rab32 interacts with SNX6 and affects retromer-dependent Golgi trafficking
Abstract
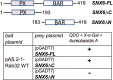
The Rab family of small GTPases regulate various aspects of cellular dynamics in eukaryotic cells. Membrane trafficking has emerged as central to the functions of leucine-rich repeat kinase 2 (LRRK2), which is associated with inherited and sporadic forms of Parkinson's disease (PD). Rabs act as both regulators of the catalytic activity and targets for serine/threonine phosphorylation by LRRK2. Rab32, Rab38 and Rab29 have been shown to regulate LRRK2 sub-cellular localization through direct interactions. Recently, Rab29 was shown to escort LRRK2 to the Golgi apparatus and activate the phosphorylation of Rab8 and Rab10. Rab32 is linked to multiple cellular functions including endosomal trafficking, mitochondrial dynamics, and melanosome biogenesis. A missense mutation in Rab32 has also recently been linked to PD. Here, we demonstrate that Rab32 directly interacts with sorting nexin 6 (SNX6). SNX6 is a transient subunit of the retromer, an endosome-Golgi retrieval complex whose Vps35 subunit is strongly associated with PD. We could further show that localization of cation-independent mannose-6-phosphate receptors, which are recycled to the trans-Golgi network (TGN) by the retromer, was affected by both Rab32 and SNX6. These data imply that Rab32 is linked to SNX6/retromer trafficking at the Golgi, and also suggests a possible connection between the retromer and Rab32 in the trafficking and biological functions of LRRK2.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

