Predictors of the effectiveness of an early medication change strategy in patients with major depressive disorder
- PMID: 30642308
- PMCID: PMC6332626
- DOI: 10.1186/s12888-019-2014-x
Predictors of the effectiveness of an early medication change strategy in patients with major depressive disorder
Abstract
Background: Patients with Major Depressive Disorder (MDD) who are non-improvers after two weeks of antidepressant treatment have a high risk of treatment failure. Recently, we did not find differences in outcomes in non-improvers randomized to an early medication change (EMC) strategy compared to treatment as usual (TAU). This secondary analysis investigated possible predictors of higher remission rates in the EMC strategy.
Methods: Of 192 non-improvers (i.e. decrease of ≤20% on the HAMD-17 depression scale) after a two-week treatment with escitalopram, n = 97 were randomized to EMC (immediate switch to high doses of venlafaxine XR) and n = 95 to TAU (continued escitalopram until day 28 with non-responders switched to venlafaxine XR). We first analyzed patient characteristics, psychopathological features and subtypes of MDD by logistic regression analyses as possible predictors of remission rates. In a second investigation, we analyzed the predictors, which showed a significant association in the first analysis before Bonferroni-Holm correction by chi-squared tests separated for treatment groups. All analyses were corrected by Bonferroni-Holm method.
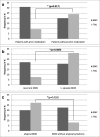
Results: The first analyses yielded no statistically significant results after correction for multiple testing. In the second analyses, however, patients with prior medication at study entry showed higher remission rates in EMC than in TAU (24.2% versus 8.6%, p = 0.017; Bonferroni-Holm corrected significance level: p = 0.025.). Furthermore, patients with a recurrent course of MDD benefited less from treatment as usual (p = 0.009; Bonferroni-Holm corrected significance level: p = 0.025). Age, sex, age of onset, psychiatric or somatic comorbidities, and other subtypes of MDD did not predict remission rates.
Conclusions: Although in our first analysis we found statistically non-significant results, the second analysis showed significant differences in remission rates between patients with or without previous medication and in patients with recurrent MDD or the first depressive episode. It would therefore be valuable to examine in larger and prospective studies whether remission rates can be increased by quick escalation of treatment in certain subgroups of patients. Promising subgroups to be tested are patients who were previously medicated, and who show a recurrent course of MDD.
Trial registration: clinicaltrials.gov Identifier: NCT00974155 . Registered at the 10th of September 2009. Retrospectively registered.
Keywords: Antidepressant; Early improvement; Major depressive disorder; Predictor; Treatment outcome.
Conflict of interest statement
Ethics approval and consent to participate
The trial was approved in July 2009 by the ethics committee at the Landesärztekammer Rheinland-Pfalz (code: 837.211.09 (6717)) and the German Federal Institute for Drugs and Medical Devices (BfArM) approved the trial protocol. All study participants provided written informed consent at enrollment into the protocol treatment. The study was conducted in accordance with the Helsinki Declaration and is compliant with the CONSORT guidelines.
Consent for publication
Not applicable
Competing interests
AT has received consultancy fees from Janssen and Novartis. All other authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Randomized controlled study of early medication change for non-improvers to antidepressant therapy in major depression--The EMC trial.Eur Neuropsychopharmacol. 2016 Apr;26(4):705-16. doi: 10.1016/j.euroneuro.2016.02.003. Epub 2016 Feb 9. Eur Neuropsychopharmacol. 2016. PMID: 26899588 Clinical Trial.
-
Rationale and design of the randomised clinical trial comparing early medication change (EMC) strategy with treatment as usual (TAU) in patients with major depressive disorder--the EMC trial.Trials. 2010 Feb 26;11:21. doi: 10.1186/1745-6215-11-21. Trials. 2010. PMID: 20187947 Free PMC article. Clinical Trial.
-
Novel Augmentation Strategies in Major Depression.Dan Med J. 2017 Apr;64(4):B5338. Dan Med J. 2017. PMID: 28385173 Review.
-
Does early-onset chronic or recurrent major depression impact outcomes with antidepressant medications? A CO-MED trial report.Psychol Med. 2013 May;43(5):945-60. doi: 10.1017/S0033291712001742. Epub 2012 Dec 11. Psychol Med. 2013. PMID: 23228340 Clinical Trial.
-
Early switching strategies in antidepressant non-responders: current evidence and future research directions.CNS Drugs. 2014 Jul;28(7):601-9. doi: 10.1007/s40263-014-0171-5. CNS Drugs. 2014. PMID: 24831418 Review.
Cited by
-
Concordance of the treatment patterns for major depressive disorders between the Canadian Network for Mood and Anxiety Treatments (CANMAT) algorithm and real-world practice in China.Front Pharmacol. 2022 Aug 31;13:954973. doi: 10.3389/fphar.2022.954973. eCollection 2022. Front Pharmacol. 2022. PMID: 36120331 Free PMC article.
References
-
- Global Burden of Disease Study C Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386:743–800. doi: 10.1016/S0140-6736(15)60692-4. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

