Functional and morphologic study of retinal hypoperfusion injury induced by bilateral common carotid artery occlusion in rats
- PMID: 30643163
- PMCID: PMC6331588
- DOI: 10.1038/s41598-018-36400-5
Functional and morphologic study of retinal hypoperfusion injury induced by bilateral common carotid artery occlusion in rats
Abstract
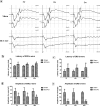
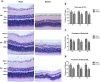
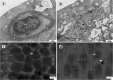
Retinal hypoperfusion injury is the pathophysiologic basis of ocular ischemic syndrome (OIS) which often leads to severe visual loss. In this study, we aimed to establish a rat model of retinal chronic hypoperfusion by bilateral common carotid artery occlusion (BCCAO) and observe changes in the retinal function and morphology. We found that model rats showed retinal arteriosclerosis, slight dilated retinal vein, small hemangiomas, hemorrhages, vascular segmental filling, and nonperfused areas after 2 weeks of BCCAO. In the model rats, the retinal circulation time was significantly prolonged by fluorescein fundus angiography (FFA), the latency of a and b waves was delayed and the amplitude was decreased significantly at each time point by electroretinogram (ERG), and the perfusion of the eyes continued to reduced. Morphologic and ultrastructural changes covered that the retinal ganglion cells (RGCs) presented obvious apoptosis and the thickness in the retinal layers were significantly thinner. Collectively, these findings suggested that BCCAO induced retinal hypoperfusion injury in the model rats, thus providing an ideal animal model for the study of OIS.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Collapsin Response Mediator Protein-2-induced Retinal Ischemic Injury in a Novel Mice Model of Ocular Ischemia Syndrome.Chin Med J (Engl). 2017 Jun 5;130(11):1342-1351. doi: 10.4103/0366-6999.206340. Chin Med J (Engl). 2017. PMID: 28524835 Free PMC article.
-
Chronic bilateral common carotid artery occlusion: a model for ocular ischemic syndrome in the rat.Graefes Arch Clin Exp Ophthalmol. 2006 Feb;244(2):199-204. doi: 10.1007/s00417-005-0006-7. Epub 2005 Jun 28. Graefes Arch Clin Exp Ophthalmol. 2006. PMID: 15983810
-
Ligation of the pterygopalatine and external carotid arteries induces ischemic damage in the murine retina.Invest Ophthalmol Vis Sci. 2011 Dec 28;52(13):9710-20. doi: 10.1167/iovs.11-8160. Invest Ophthalmol Vis Sci. 2011. PMID: 22110078
-
[Nonarteritic ischemic optic neuropathy animal model and its treatment applications].Nippon Ganka Gakkai Zasshi. 2014 Apr;118(4):331-61. Nippon Ganka Gakkai Zasshi. 2014. PMID: 24864434 Review. Japanese.
-
[Ocular changes in internal carotid artery diseases caused by artheriosclerosis].Pol Merkur Lekarski. 2008 Feb;24(140):170-2. Pol Merkur Lekarski. 2008. PMID: 18634278 Review. Polish.
Cited by
-
Changes of retinal structure and function in patients with internal carotid artery stenosis.BMC Ophthalmol. 2022 Mar 15;22(1):123. doi: 10.1186/s12886-022-02345-7. BMC Ophthalmol. 2022. PMID: 35287632 Free PMC article.
-
Fenofibrate Protects against Retinal Dysfunction in a Murine Model of Common Carotid Artery Occlusion-Induced Ocular Ischemia.Pharmaceuticals (Basel). 2021 Mar 7;14(3):223. doi: 10.3390/ph14030223. Pharmaceuticals (Basel). 2021. PMID: 33799938 Free PMC article.
-
Neuroprotective Effects of Human Adipose-Derived Mesenchymal Stem Cells in Oxygen-Induced Retinopathy.Cell Transplant. 2023 Jan-Dec;32:9636897231213309. doi: 10.1177/09636897231213309. Cell Transplant. 2023. PMID: 38018498 Free PMC article.
-
Relation of Retinal Oxygen Measures to Electrophysiology and Survival Indicators after Permanent, Incomplete Ischemia in Rats.Transl Stroke Res. 2020 Dec;11(6):1273-1286. doi: 10.1007/s12975-020-00799-9. Epub 2020 Mar 24. Transl Stroke Res. 2020. PMID: 32207038 Free PMC article.
-
Lacidipine, thiamine pyrophosphate and their combination on the ocular ischemic syndrome induced by bilateral common carotid artery ligation.Int J Ophthalmol. 2024 May 18;17(5):815-821. doi: 10.18240/ijo.2024.05.04. eCollection 2024. Int J Ophthalmol. 2024. PMID: 38766328 Free PMC article.
References
-
- Sivalingam A, Brown GC, Magargal LE. The ocular ischemic syndrome. III. Visual prognosis and the effect of treatment. Int Ophthalmol. 1991;15:15–20. - PubMed
-
- Sharma, S. & Brown, G. C. Chapter 84-Ocular Ischemic Syndrome. Retina 1491–1502 (2006).
-
- Zhang, H. R. Ocular microcirculation and related diseases. Beijing Medical University-China Union Medical University United Press (1993).
Publication types
MeSH terms
Grants and funding
- 81574029/National Natural Science Foundation of China (National Science Foundation of China)/International
- 81574029/National Natural Science Foundation of China (National Science Foundation of China)/International
- 81574029/National Natural Science Foundation of China (National Science Foundation of China)/International
- 81574029/National Natural Science Foundation of China (National Science Foundation of China)/International
LinkOut - more resources
Full Text Sources
Medical

