Primary systemic therapy in HER2-positive operable breast cancer using trastuzumab and chemotherapy: efficacy data, cardiotoxicity and long-term follow-up in 142 patients diagnosed from 2005 to 2016 at a single institution
- PMID: 30643452
- PMCID: PMC6311333
- DOI: 10.2147/BCTT.S179750
Primary systemic therapy in HER2-positive operable breast cancer using trastuzumab and chemotherapy: efficacy data, cardiotoxicity and long-term follow-up in 142 patients diagnosed from 2005 to 2016 at a single institution
Abstract
Objective: The aim of this study was to evaluate the efficacy, cardiotoxicity profile and long-term benefits of neoadjuvant therapy in human epidermal growth factor receptor 2-positive operable breast cancer patients.
Patients and methods: A total of 142 patients diagnosed from 2005 to 2016 were included in the study. The treatment consisted of a sequential regimen of taxanes and anthracyclines plus trastuzumab. The clinical and pathological responses were evaluated and correlated with clinical and biological factors. The cardiotoxicity profile and long-term benefits were analyzed.
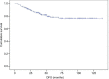
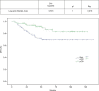
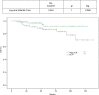
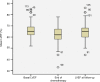
Results: The median age was 49 years, and 4%, 69% and 27% of patients had stage I, II and III breast cancer, respectively, while 10% had inflammatory breast cancer at diagnosis. Hormone receptor (HR) status was negative in 43%, and 62% had grade III breast cancer. The clinical complete response rate was 49% and 63% as assessed using ultrasound and magnetic resonance imaging, respectively, and this allowed a high rate of conservative surgery (66%). The pathological complete response (pCR) rate was 52%, and it was higher in HR-negative (64%) patients than in HR-positive (41%) patients and in grade III breast cancer (53%) patients than in grade I-II breast cancer (45%) patients. Patients who achieved pCR had longer disease-free survival and a trend toward improved overall survival. A total of 2% of patients showed a 10% decrease in left ventricular ejection fraction to <50% during treatment. All patients except one recovered after discontinuation of trastuzumab.
Conclusion: A sequential regimen of taxanes and anthracyclines plus trastuzumab was effective, with high pCR rates and long-term benefit, and had a very good cardiotoxicity profile.
Keywords: HER2-positive breast cancer; cardiotoxicity; neoadjuvant therapy; pathological complete response; survival.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures


References
-
- Hortobagyi GN, Blumenschein GR, Spanos W, et al. Multimodal treatment of locoregionally advanced breast cancer. Cancer. 1983;51(5):763–768. - PubMed
-
- Hortobagyi GN, Ames FC, Buzdar AU, et al. Management of stage III primary breast cancer with primary chemotherapy, surgery, and radiation therapy. Cancer. 1988;62(12):2507–2516. - PubMed
-
- Bear HD. Indications for neoadjuvant chemotherapy for breast cancer. Semin Oncol. 1998;25(2 Suppl 3):3–12. - PubMed
-
- Rastogi P, Anderson SJ, Bear HD, et al. Preoperative chemotherapy: updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27. J Clin Oncol. 2008;26(5):778–785. - PubMed
-
- Mauri D, Pavlidis N, Ioannidis JP. Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst. 2005;97(3):188–194. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

