Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape
- PMID: 30646912
- PMCID: PMC6332843
- DOI: 10.1186/s12943-018-0928-4
Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape
Abstract
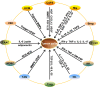
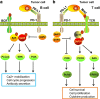
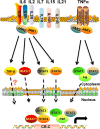
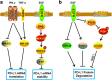
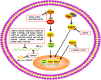
Tumor immune escape is an important strategy of tumor survival. There are many mechanisms of tumor immune escape, including immunosuppression, which has become a research hotspot in recent years. The programmed death ligand-1/programmed death-1 (PD-L1/PD-1) signaling pathway is an important component of tumor immunosuppression, which can inhibit the activation of T lymphocytes and enhance the immune tolerance of tumor cells, thereby achieving tumor immune escape. Therefore, targeting the PD-L1/PD-1 pathway is an attractive strategy for cancer treatment; however, the therapeutic effectiveness of PD-L1/PD-1 remains poor. This situation requires gaining a deeper understanding of the complex and varied molecular mechanisms and factors driving the expression and activation of the PD-L1/PD-1 signaling pathway. In this review, we summarize the regulation mechanisms of the PD-L1/PD-1 signaling pathway in the tumor microenvironment and their roles in mediating tumor escape. Overall, the evidence accumulated to date suggests that induction of PD-L1 by inflammatory factors in the tumor microenvironment may be one of the most important factors affecting the therapeutic efficiency of PD-L1/PD-1 blocking.
Keywords: Inflammatory factor; PD-1; PD-L1; Tumor immune escape; Tumor microenvironment.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- Jiang XJ, Wang J, Deng XY, Li XL, Li XY, Zeng ZY, et al. Immunotherapy targeted to immune checkpoint: a revolutionary breakthrough in cancer therapy. Prog Biochem Biophys. 2018;45(11):1178–86.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

