NETest Liquid Biopsy Is Diagnostic of Lung Neuroendocrine Tumors and Identifies Progressive Disease
- PMID: 30654372
- PMCID: PMC7472425
- DOI: 10.1159/000497037
NETest Liquid Biopsy Is Diagnostic of Lung Neuroendocrine Tumors and Identifies Progressive Disease
Abstract
Background: There are no effective biomarkers for the management of bronchopulmonary carcinoids (BPC). We examined the utility of a neuroendocrine multigene transcript "liquid biopsy" (NETest) in BPC for diagnosis and monitoring of the disease status.
Aim: To independently validate the utility of the NETest in diagnosis and management of BPC in a multicenter, multinational, blinded study.
Material and methods: The study cohorts assessed were BPC (n = 99), healthy controls (n = 102), other lung neoplasia (n = 101) including adenocarcinomas (ACC) (n = 41), squamous cell carcinomas (SCC) (n = 37), small-cell lung cancer (SCLC) (n = 16), large-cell neuroendocrine carcinoma (LCNEC) (n = 7), and idiopathic pulmonary fibrosis (IPF) (n = 50). BPC were histologically classified as typical (TC) (n = 62) and atypical carcinoids (AC) (n = 37). BPC disease status determination was based on imaging and RECIST 1.1. NETest diagnostic metrics and disease status accuracy were evaluated. The upper limit of normal (NETest) was 20. Twenty matched tissue-blood pairs were also evaluated. Data are means ± SD.
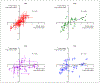
Results: NETest levels were significantly increased in BPC (45 ± 25) versus controls (9 ± 8; p < 0.0001). The area under the ROC curve was 0.96 ± 0.01. Accuracy, sensitivity, and specificity were: 92, 84, and 100%. NETest was also elevated in SCLC (42 ± 32) and LCNEC (28 ± 7). NETest accurately distinguished progressive (61 ± 26) from stable disease (35.5 ± 18; p < 0.0001). In BPC, NETest levels were elevated in metastatic disease irrespective of histology (AC: p < 0.02; TC: p = 0.0006). In nonendocrine lung cancers, ACC (18 ± 21) and SCC (12 ± 11) and benign disease (IPF) (18 ± 25) levels were significantly lower compared to BPC level (p < 0.001). Significant correlations were evident between paired tumor and blood samples for BPC (R: 0.83, p < 0.0001) and SCLC (R: 0.68) but not for SCC and ACC (R: 0.25-0.31).
Conclusions: Elevated -NETest levels are indicative of lung neuroendocrine neoplasia. NETest levels correlate with tumor tissue and imaging and accurately define clinical progression.
Keywords: Biomarker; Blood; Bronchopulmonary carcinoid; Lung cancer; NETest; Neuroendocrine tumor; PCR; Progression; Transcript.
© 2019 S. Karger AG, Basel.
Conflict of interest statement
Disclosure Statement
There are no conflicts of interest that could be perceived as prejudicing the impartiality of the research reported.
Figures

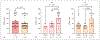



References
-
- Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2008. July; 113(1): 5–21. - PubMed
-
- Caplin ME, Baudin E, Ferolla P, Filosso P, Garcia-Yuste M, Lim E, et al.; ENETS consensus conference participants. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015. August; 26(8): 1604–20. - PubMed
-
- Filosso PL, Guerrera F, Thomas P, Brunelli A, Lim E, Garcia-Yuste M, et al.; European Society of Thoracic Surgeons Neuroendocrine Tumors of the Lung Working Group. Management of bronchial carcinoids: international practice survey among the European Society of Thoracic Surgeons. Future Oncol. 2016. September; 12(17): 1985–99. - PubMed
-
- Oberg K, Couvelard A, Delle Fave G, Gross D, Grossman A, Jensen RT, et al.; Antibes Consensus Conference participants. ENETS Consensus Guidelines for Standard of Care in Neuroendocrine Tumours: biochemical markers. Neuroendocrinology. 2017;105(3):201–11. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

