The Effect of iPS-Derived Neural Progenitors Seeded on Laminin-Coated pHEMA-MOETACl Hydrogel with Dual Porosity in a Rat Model of Chronic Spinal Cord Injury
- PMID: 30654639
- PMCID: PMC6628561
- DOI: 10.1177/0963689718823705
The Effect of iPS-Derived Neural Progenitors Seeded on Laminin-Coated pHEMA-MOETACl Hydrogel with Dual Porosity in a Rat Model of Chronic Spinal Cord Injury
Abstract
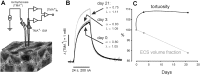
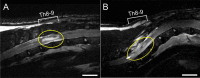
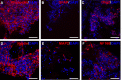
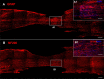
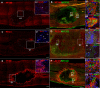
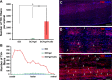
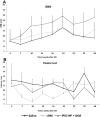
Spinal cord injury (SCI), is a devastating condition leading to the loss of locomotor and sensory function below the injured segment. Despite some progress in acute SCI treatment using stem cells and biomaterials, chronic SCI remains to be addressed. We have assessed the use of laminin-coated hydrogel with dual porosity, seeded with induced pluripotent stem cell-derived neural progenitors (iPSC-NPs), in a rat model of chronic SCI. iPSC-NPs cultured for 3 weeks in hydrogel in vitro were positive for nestin, glial fibrillary acidic protein (GFAP) and microtubule-associated protein 2 (MAP2). These cell-polymer constructs were implanted into a balloon compression lesion, 5 weeks after lesion induction. Animals were behaviorally tested, and spinal cord tissue was immunohistochemically analyzed 28 weeks after SCI. The implanted iPSC-NPs survived in the scaffold for the entire experimental period. Host axons, astrocytes and blood vessels grew into the implant and an increased sprouting of host TH+ fibers was observed in the lesion vicinity. The implantation of iPSC-NP-LHM cell-polymer construct into the chronic SCI led to the integration of material into the injured spinal cord, reduced cavitation and supported the iPSC-NPs survival, but did not result in a statistically significant improvement of locomotor recovery.
Keywords: Chronic spinal cord injury; HEMA hydrogel; human induced pluripotent stem cells; laminin; neural progenitors; surface charge.
Conflict of interest statement
Figures

Similar articles
-
Beneficial Effect of Human Induced Pluripotent Stem Cell-Derived Neural Precursors in Spinal Cord Injury Repair.Cell Transplant. 2015;24(9):1781-97. doi: 10.3727/096368914X684042. Epub 2014 Aug 19. Cell Transplant. 2015. PMID: 25259685
-
Effects of the Post-Spinal Cord Injury Microenvironment on the Differentiation Capacity of Human Neural Stem Cells Derived from Induced Pluripotent Stem Cells.Cell Transplant. 2016 Oct;25(10):1833-1852. doi: 10.3727/096368916X691312. Cell Transplant. 2016. PMID: 27075820
-
HPMA-RGD hydrogels seeded with mesenchymal stem cells improve functional outcome in chronic spinal cord injury.Stem Cells Dev. 2010 Oct;19(10):1535-46. doi: 10.1089/scd.2009.0378. Stem Cells Dev. 2010. PMID: 20053128
-
Bone marrow stem cells and polymer hydrogels--two strategies for spinal cord injury repair.Cell Mol Neurobiol. 2006 Oct-Nov;26(7-8):1113-29. doi: 10.1007/s10571-006-9007-2. Epub 2006 Apr 22. Cell Mol Neurobiol. 2006. PMID: 16633897 Free PMC article. Review.
-
Progress in the Use of Induced Pluripotent Stem Cell-Derived Neural Cells for Traumatic Spinal Cord Injuries in Animal Populations: Meta-Analysis and Review.Stem Cells Transl Med. 2019 Jul;8(7):681-693. doi: 10.1002/sctm.18-0225. Epub 2019 Mar 22. Stem Cells Transl Med. 2019. PMID: 30903654 Free PMC article.
Cited by
-
Hydrogels in Spinal Cord Injury Repair: A Review.Front Bioeng Biotechnol. 2022 Jun 21;10:931800. doi: 10.3389/fbioe.2022.931800. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35800332 Free PMC article. Review.
-
Combined application of neural stem/progenitor cells and scaffolds on locomotion recovery following spinal cord injury in rodents: a systematic review and meta-analysis.Neurosurg Rev. 2022 Dec;45(6):3469-3488. doi: 10.1007/s10143-022-01859-4. Epub 2022 Sep 17. Neurosurg Rev. 2022. PMID: 36114918
-
Cost-Effective Cosmetic-Grade Hyaluronan Hydrogels for ReNcell VM Human Neural Stem Cell Culture.Biomolecules. 2019 Sep 20;9(10):515. doi: 10.3390/biom9100515. Biomolecules. 2019. PMID: 31547190 Free PMC article.
-
Stem Cell Therapies for Restorative Treatments of Central Nervous System Ischemia-Reperfusion Injury.Cell Mol Neurobiol. 2023 Mar;43(2):491-510. doi: 10.1007/s10571-022-01204-9. Epub 2022 Feb 7. Cell Mol Neurobiol. 2023. PMID: 35129759 Free PMC article. Review.
-
Probing of New Polymer-Based Microcapsules for Islet Cell Immunoisolation.Polymers (Basel). 2024 Aug 30;16(17):2479. doi: 10.3390/polym16172479. Polymers (Basel). 2024. PMID: 39274113 Free PMC article.
References
-
- Sekhon LH, Fehlings MG. Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine (Phila Pa 1976). 2001;26(24 Suppl):S2–S12. - PubMed
-
- Fawcett JW. The glial response to injury and its role in the inhibition of CNS repair. Adv Exp Med Biol. 2006;557:11–24. - PubMed
-
- Schwab ME. Increasing plasticity and functional recovery of the lesioned spinal cord. Prog Brain Res. 2002;137:351–359. - PubMed
-
- Amemori T, Jendelová P, Ruzickova K, Arboleda D, Syková E. Co-transplantation of olfactory ensheathing glia and mesenchymal stromal cells does not have synergistic effects after spinal cord injury in the rat. Cytotherapy. 2010;12(2):212–225. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

