Development and Validation of the Real-World Progression in Diabetes (RAPIDS) Model
- PMID: 30654704
- PMCID: PMC6624067
- DOI: 10.1177/0272989X18817521
Development and Validation of the Real-World Progression in Diabetes (RAPIDS) Model
Abstract
Introduction: To develop and validate the first real-world data-based type 2 diabetes progression model (RAPIDS) employing econometric techniques that can study the comparative effects of complex dynamic patterns of glucose-lowering drug use.
Methods: The US Department of Veterans Affairs (VA) electronic medical record and claims databases were used to identify over 500,000 diabetes patients in 2003 with up to 9-year follow-up. The RAPIDS model contains interdependent first-order Markov processes over quarters for each of the micro- and macrovascular events, hypoglycemia, and death, as well as predictive models for 8 biomarker levels. Model parameters varied by static demographic factors and dynamic factors, such as age, duration of diabetes, 13 possible glucose-lowering treatment combinations, any blood pressure and any cholesterol-lowering medications, and cardiovascular history. To illustrate model capabilities, a simple comparative study was set up to compare observed treatment use patterns to alternate patterns if perfect adherence is assumed following initiating the use of any of these medications.
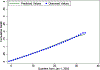
Results: Data were randomly split into 307,288, 105,195, and 105,081 patients to perform estimation, out-of-sample calibration, and validation, respectively. Model predictions in the validation sample closely aligned with the observed longitudinal trajectory of biomarkers and outcomes. Perfect adherence among initiators increased proportion of days covered by only 6 months. Most of this increase came from increased adherence to monotherapies and did not lead to meaningful changes in any of the outcomes over the 9-year period.
Conclusion: Future value of increasing medication adherence among VA patients with diabetes may lie among those who never initiate treatment or are late in initiating treatment. The first-of-its-kind real-world data-based model has the potential to carry out many complex comparative-effectiveness research (CER) studies of dynamic glucose-lowering drug regimens.
Keywords: comparative effectiveness research; diabetes; dynamic patterns; glucose-lowering; model; real-world data.
Figures




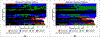
References
-
- Turner RC, Cull CA, Frighi V, Holman RR. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA 1999;281(21):2005–12. - PubMed
-
- Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA 2007;298(2):194–206. - PubMed
-
- Nathan DM, Buse JB, Davidson MB, Ferrannini E, Holman RR, Sherwin R, et al. Management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: update regarding thiazolidinediones: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2008;31(1):173–5. - PubMed
-
- Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358(24):2560–72. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

