Efficacy and safety of exogenous ketone bodies for preventive treatment of migraine: A study protocol for a single-centred, randomised, placebo-controlled, double-blind crossover trial
- PMID: 30654835
- PMCID: PMC6337840
- DOI: 10.1186/s13063-018-3120-7
Efficacy and safety of exogenous ketone bodies for preventive treatment of migraine: A study protocol for a single-centred, randomised, placebo-controlled, double-blind crossover trial
Abstract
Background: Currently available prophylactic migraine treatment options are limited and are associated with many, often intolerable, side-effects. Various lines of research suggest that abnormalities in energy metabolism are likely to be part of migraine pathophysiology. Previously, a ketogenic diet (KD) has been reported to lead to a drastic reduction in migraine frequency. An alternative method to a strict KD is inducing a mild nutritional ketosis (0.4-2 mmol/l) with exogenous ketogenic substances. The aim of this randomised, placebo-controlled, double-blind, crossover, single-centre trial is to demonstrate safety and superiority of beta-hydroxybutyrate (βHB) in mineral salt form over placebo in migraine prevention.
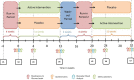
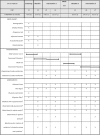
Methods/design: Forty-five episodic migraineurs (5-14 migraine days/months), with or without aura, aged between 18 and 65 years, will be recruited at headache clinics in Switzerland, Germany and Austria and via Internet announcements. After a 4-week baseline period, patients will be randomly allocated to one of the two trial arms and receive either the βHB mineral salt or placebo for 12 weeks. This will be followed by a 4-week wash-out period, a subsequent second baseline period and, finally, another 12-week intervention with the alternative treatment. Co-medication with triptans (10 days per months) or analgesics (14 days per months) is permitted. The primary outcome is the mean change from baseline in the number of migraine days (meeting International Classification of Headache Disorders version 3 criteria) during the last 4 weeks of intervention compared to placebo. Secondary endpoints include mean changes in headache days of any severity, acute migraine medication use, migraine intensity and migraine and headache-related disability. Exploratory outcomes are (in addition to routine laboratory analysis) genetic profiling and expression analysis, oxidative and nitrosative stress, as well as serum cytokine analysis, and blood βHB and glucose analysis (pharmacokinetics).
Discussion: A crossover design was chosen as it greatly improves statistical power and participation rates, without increasing costs. To our knowledge this is the first RCT using βHB salts worldwide. If proven effective and safe, βHB might not only offer a new prophylactic treatment option for migraine patients, but might additionally pave the way for clinical trials assessing its use in related diseases.
Trial registration: ClinicalTrials.gov, NCT03132233 . Registered on 27 April 2017.
Keywords: 3-Hydroxybutyrate; Beta-hydroxybutyrate; Clinical trial; Crossover; Exogenous ketone bodies; Ketosis; Migraine; Migraine prevention; Placebo-controlled; Randomised controlled trial.
Conflict of interest statement
Ethics approval and consent to participate
Ethics approval has been obtained from the local Ethics Committee (EKNZ 2015-304) and the National Swiss Drug Agency (2016DR2109).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

