A phase 1/2 ascending dose study and open-label extension study of voxelotor in patients with sickle cell disease
- PMID: 30655275
- PMCID: PMC6484388
- DOI: 10.1182/blood-2018-08-868893
A phase 1/2 ascending dose study and open-label extension study of voxelotor in patients with sickle cell disease
Abstract
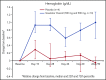
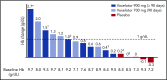
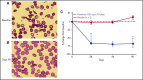
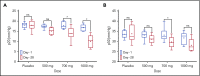
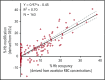
New treatments directly targeting polymerization of sickle hemoglobin (HbS), the proximate event in the pathophysiology of sickle cell disease (SCD), are needed to address the severe morbidity and early mortality associated with the disease. Voxelotor (GBT440) is a first-in-class oral therapy specifically developed to treat SCD by modulating the affinity of hemoglobin (Hb) for oxygen, thus inhibiting HbS polymerization and downstream adverse effects of hemolytic anemia and vaso-occlusion. GBT440-001 was a phase 1/2 randomized, double-blind, placebo-controlled, single and multiple ascending dose study of voxelotor in adult healthy volunteers and patients with SCD, followed by a single-arm, open-label extension study. This report describes results of voxelotor (500-1000 mg per day) in patients with sickle cell anemia. The study evaluated the safety, tolerability, pharmacokinetic, and pharmacodynamic properties of voxelotor and established proof of concept by improving clinical measures of anemia, hemolysis, and sickling. Thirty-eight patients with SCD received 28 days of voxelotor 500, 700, or 1000 mg per day or placebo; 16 patients received 90 days of voxelotor 700 or 900 mg per day or placebo. Four patients from the 90-day cohort were subsequently enrolled in an extension study and treated with voxelotor 900 mg per day for 6 months. All patients who received multiple doses of voxelotor for ≥28 days experienced hematologic improvements including increased Hb and reduction in hemolysis and percentage of sickled red cells, supporting the potential of voxelotor to serve as a disease-modifying therapy for SCD. Voxelotor was well tolerated with no treatment-related serious adverse events and no evidence of tissue hypoxia. These trials were registered at www.clinicaltrials.gov as #NCT02285088 and #NCT03041909.
© 2019 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: J.H. is a consultant/advisor for Novartis; Global Blood Therapeutics, Inc; Bluebird Bio; and Terumo BCT. P.T. is a consultant/advisor for Apopharma; Bluebird Bio; Global Blood Therapeutics, Inc; Novartis; and Terumo. D.M.L. is a consultant/advisor for Agios, Cerus, and Novartis. J.P. received research funding from Novartis; was a consultant/advisor for Novartis, Shire, and Celgene; and received honoraria from Novartis and Celgene. M.A. is a consultant for, and receives honoraria from, Novartis. T.M. is an employee of, and owns shares of, IQVIA. D.D.G. is an independent consultant. K.D., A.H., M.P., V.S., S.D., M.T., and J.L.-G. are employees of, and have equity ownership in, Global Blood Therapeutics, Inc. N.L. is a consultant clinical site liaison for Global Blood Therapeutics, Inc. C.J.H. declares no competing financial interests.
Figures


Comment in
-
Primary polymerization prevention.Blood. 2019 Apr 25;133(17):1797-1798. doi: 10.1182/blood-2019-02-898767. Blood. 2019. PMID: 31023742 No abstract available.
Similar articles
-
Pharmacokinetics and pharmacodynamics of voxelotor (GBT440) in healthy adults and patients with sickle cell disease.Br J Clin Pharmacol. 2019 Jun;85(6):1290-1302. doi: 10.1111/bcp.13896. Epub 2019 Mar 31. Br J Clin Pharmacol. 2019. PMID: 30743314 Free PMC article. Clinical Trial.
-
Voxelotor: A Novel Treatment for Sickle Cell Disease.Ann Pharmacother. 2021 Feb;55(2):240-245. doi: 10.1177/1060028020943059. Epub 2020 Jul 16. Ann Pharmacother. 2021. PMID: 32674605 Review.
-
Voxelotor in adolescents and adults with sickle cell disease (HOPE): long-term follow-up results of an international, randomised, double-blind, placebo-controlled, phase 3 trial.Lancet Haematol. 2021 May;8(5):e323-e333. doi: 10.1016/S2352-3026(21)00059-4. Epub 2021 Apr 7. Lancet Haematol. 2021. PMID: 33838113 Clinical Trial.
-
Systematic Review of Voxelotor: A First-in-Class Sickle Hemoglobin Polymerization Inhibitor for Management of Sickle Cell Disease.Pharmacotherapy. 2020 Jun;40(6):525-534. doi: 10.1002/phar.2405. Epub 2020 May 19. Pharmacotherapy. 2020. PMID: 32343424
-
A Phase 3 Randomized Trial of Voxelotor in Sickle Cell Disease.N Engl J Med. 2019 Aug 8;381(6):509-519. doi: 10.1056/NEJMoa1903212. Epub 2019 Jun 14. N Engl J Med. 2019. PMID: 31199090 Clinical Trial.
Cited by
-
Voxelotor: alteration of sickle cell disease pathophysiology by a first-in-class polymerization inhibitor.Ther Adv Hematol. 2021 Mar 19;12:20406207211001136. doi: 10.1177/20406207211001136. eCollection 2021. Ther Adv Hematol. 2021. PMID: 33796238 Free PMC article. Review.
-
Voxelotor: A Ray of Hope for Sickle Disease.Cureus. 2020 Feb 26;12(2):e7105. doi: 10.7759/cureus.7105. Cureus. 2020. PMID: 32257653 Free PMC article. Review.
-
Increased hemoglobin affinity for oxygen with GBT1118 improves hypoxia tolerance in sickle cell mice.Am J Physiol Heart Circ Physiol. 2021 Aug 1;321(2):H400-H411. doi: 10.1152/ajpheart.00048.2021. Epub 2021 Jul 2. Am J Physiol Heart Circ Physiol. 2021. PMID: 34213392 Free PMC article.
-
Emerging disease-modifying therapies for sickle cell disease.Haematologica. 2019 Sep;104(9):1710-1719. doi: 10.3324/haematol.2018.207357. Epub 2019 Aug 14. Haematologica. 2019. PMID: 31413089 Free PMC article. Review.
-
Patient perception of voxelotor treatment benefit in sickle cell disease.J Investig Med. 2022 Jun;70(5):1316-1319. doi: 10.1136/jim-2021-002215. J Investig Med. 2022. PMID: 35732337 Free PMC article.
References
-
- Piel FB, Steinberg MH, Rees DC. Sickle cell disease. N Engl J Med. 2017;376(16):1561-1573. - PubMed
-
- Bunn HF. Pathogenesis and treatment of sickle cell disease. N Engl J Med. 1997;337(11):762-769. - PubMed
-
- Powars DR, Chan LS, Hiti A, Ramicone E, Johnson C. Outcome of sickle cell anemia: a 4-decade observational study of 1056 patients. Medicine (Baltimore). 2005;84(6):363-376. - PubMed
-
- Hassell KL. Population estimates of sickle cell disease in the U.S. Am J Prev Med. 2010;38(suppl 4):S512-S521. - PubMed

