Efficacy and Safety of Delayed Prolonged-Release Tacrolimus Initiation in De Novo Hepatitis C Virus-Negative Orthotopic Liver Transplant Recipients: A Single-Center, Single-Arm, Prospective Study
- PMID: 30655498
- PMCID: PMC6346812
- DOI: 10.12659/AOT.912444
Efficacy and Safety of Delayed Prolonged-Release Tacrolimus Initiation in De Novo Hepatitis C Virus-Negative Orthotopic Liver Transplant Recipients: A Single-Center, Single-Arm, Prospective Study
Abstract
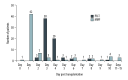
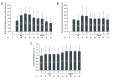
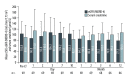
BACKGROUND Delaying initiation of tacrolimus after liver transplantation (LT) is a potential renal-sparing strategy. We assessed safety and efficacy of delayed initiation of prolonged-release tacrolimus (PR-T) in de novo LT. MATERIAL AND METHODS This was a single-center, single-arm, prospective, 12-month observational study of hepatitis C virus-negative orthotopic LT patients. Immunosuppression included PR-T (initially 0.1 or 0.2 mg/kg/day) initiated on Day 3 post LT, basiliximab (20 mg) on post-transplantation Day 0 and Day 4, and intraoperative corticosteroids (500 mg). Patients received maintenance corticosteroids and mycophenolate mofetil (MMF) according to center protocol. MMF dose was adjusted according to thrombocyte count. The primary endpoint was the estimated glomerular filtration rate (eGFR) measured using the Modification of Diet in Renal Disease 4-variable formula at 12 months. Secondary endpoints included biopsy-confirmed acute rejection (BCAR) and dialysis requirement. Adverse events were recorded. RESULTS Sixty-nine patients (mean age 55.0 years) were included. Most patients started MMF on Day 1 (60.9%) or Day 2 (10.1%), and PR-T on Day 3 (55.1%) or Day 4 (29.0%). Mean tacrolimus trough levels (ng/mL) were: Day 7, 9.5±6.3; Day 10, 9.4±5.4; Month 1, 8.0±3.1; Month 3, 7.8±3.7; Month 6, 8.0±4.1; and Month 12, 7.2±3.1. Mean 12-month eGFR was 77.2±24.5 mL/min/1.73 m2; 72.5% of patients had eGFR >60 mL/min/1.73 m² at 12 months; 89.9% had no eGFR measurements <40 mL/min/1.73 m² during the study. Renal insufficiency (any eGFR <60 mL/min/1.73 m²) was diagnosed in 27.5% of patients; one patient required dialysis. There were no BCAR episodes; the infection rate was 36.2%, and 3 patients died. Overall, 19 patients (27.5%) developed de novo diabetes mellitus, 18 patients (26.1%) had hypercholesterolemia, and 12 patients (17.4%) had hypertriglyceridemia. CONCLUSIONS Quadruple therapy with delayed administration of PR-T was well tolerated and efficacious, and was associated with acceptable renal function over 12 months.
Conflict of interest statement
LL, JF, and ER report grants from Astellas, during the conduct of the study and personal fees from Astellas outside the submitted work. CB reports grants from Astellas, during the conduct of the study and personal fees from Janssen outside the submitted work. EGV and JB report grants from Astellas, during the conduct of the study. XX reports personal fees from Gilead, outside the submitted work. AGC has no conflicts to disclose.
Figures
Similar articles
-
Incidence of Posttransplantation Diabetes Mellitus in De Novo Kidney Transplant Recipients Receiving Prolonged-Release Tacrolimus-Based Immunosuppression With 2 Different Corticosteroid Minimization Strategies: ADVANCE, A Randomized Controlled Trial.Transplantation. 2017 Aug;101(8):1924-1934. doi: 10.1097/TP.0000000000001453. Transplantation. 2017. PMID: 27547871 Free PMC article. Clinical Trial.
-
The Effect of Donor Age and Recipient Characteristics on Renal Outcomes in Patients Receiving Prolonged-Release Tacrolimus After Liver Transplantation: Post-Hoc Analyses of the DIAMOND Study.Ann Transplant. 2019 Jun 4;24:319-327. doi: 10.12659/AOT.913103. Ann Transplant. 2019. PMID: 31160549 Free PMC article.
-
ADHERE: randomized controlled trial comparing renal function in de novo kidney transplant recipients receiving prolonged-release tacrolimus plus mycophenolate mofetil or sirolimus.Transpl Int. 2017 Jan;30(1):83-95. doi: 10.1111/tri.12878. Epub 2016 Nov 28. Transpl Int. 2017. PMID: 27754567 Clinical Trial.
-
Tacrolimus once-daily formulation: in the prophylaxis of transplant rejection in renal or liver allograft recipients.Drugs. 2007;67(13):1931-43. doi: 10.2165/00003495-200767130-00012. Drugs. 2007. PMID: 17722962 Review.
-
Indications of mycophenolate mofetil in liver transplantation.Transplantation. 2005 Sep 27;80(1 Suppl):S142-6. doi: 10.1097/01.tp.0000187133.53916.8f. Transplantation. 2005. PMID: 16286893 Review.
Cited by
-
A Systematic Review of the Literature on Chronic Kidney Disease Following Liver Transplantation.Ann Transplant. 2022 May 24;27:e935170. doi: 10.12659/AOT.935170. Ann Transplant. 2022. PMID: 35607264 Free PMC article.
References
-
- Morrisey PE, Flynn ML, Lin S, et al. Medication noncompliance and its implications in transplant recipients. Drugs. 2007;67:1463–81. - PubMed
-
- Ojo AO, Held PJ, Port FK, et al. Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med. 2003;349:931–40. - PubMed
-
- Zhang W, Fung J. Limitations of current liver transplant immunosuppressive regimens: Renal considerations. Hepatobiliary Pancreat Dis Int. 2017;16:27–32. - PubMed
-
- Durand F. Hot-topic debate on kidney function: Renal-sparing approaches are beneficial. Liver Transplant. 2011;17:S43–49. - PubMed
-
- Nankivell BJ, Chapman JR, Bonovas G, Gruenewald SM. Oral cyclosporine but not tacrolimus reduces renal transplant blood flow. Transplantation. 2004;77:1457–59. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

