Apoptosis of mouse myeloma cells induced by curcumin via the Notch3-p53 signaling axis
- PMID: 30655747
- PMCID: PMC6313093
- DOI: 10.3892/ol.2018.9591
Apoptosis of mouse myeloma cells induced by curcumin via the Notch3-p53 signaling axis
Abstract
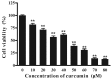
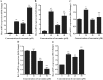
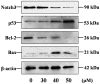
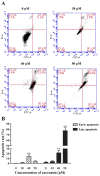
Resistance to apoptosis is a characteristic of cancer. Curcumin has become a potential anticancer drug for its pro-apoptotic effects, but the underlying mechanisms remain unclear. Furthermore, the Notch3-p53 signaling axis serves an important role in cell fate. The present study was designed to investigate the antitumor effect of curcumin by the Notch3-p53 axis in mouse myeloma P3X63Ag8 cells. The effects of curcumin on the viability of P3X63Ag8 cells were evaluated using an MTT assay. Quantitative expression of the Notch3-p53 signaling axis-associated genes was measured by reverse transcription-quantitative polymerase chain reaction, and western blot analysis was used to investigate the expression of proteins. Additionally, flow cytometry was used to measure the ratio of apoptosis. The results demonstrated that curcumin could significantly inhibit cell viability. No significant pro-apoptotic effect was observed when the concentration of curcumin was <30 µM. At 30 µM, curcumin-treated cells exhibited an apoptotic phenomenon, and the ratio of late apoptosis increased with the concentration of curcumin, and reached 28.4 and 51.8% in the medium- and high-dose groups, respectively. Curcumin inhibited the expression of Notch3, while the middle- and high-dose groups promoted p53. The expression of Notch3-responsive genes Hes family BHLH transcription factor 1 and Hes-related family transcription factor with YRPW motif 1 were notably promoted. Curcumin treatment significantly downregulated B-cell lymphoma-2 (Bcl-2) at the mRNA and protein levels, but upregulated Bcl-2-associated X. These data indicated that curcumin exhibited antitumor effects in mouse myeloma cells with induction of apoptosis by affecting the Notch3-p53 signaling axis.
Keywords: Notch3; P3X63Ag8 cell; apoptosis; curcumin; p53.
Figures
References
-
- Zhang X, Wang R, Chen G, Dejean L, Chen QH. The Effects of curcumin-based compounds on proliferation and cell death in cervical cancer cells. Anticancer Res. 2015;35:5293–5298. - PubMed
-
- Ting CY, Wang HE, Yu CC, Liu HC, Liu YC, Chiang IT. Curcumin triggers DNA damage and inhibits expression of DNA repair proteins in human lung cancer cells. Anticancer Res. 2015;35:3867–3873. - PubMed
-
- Mukhopadhyay A, Banerjee S, Stafford LJ, Xia C, Liu M, Aggarwal BB. Curcumin-induced suppression of cell proliferation correlates with down-regulation of cyclin D1 expression and CDK4-mediated retinoblastoma protein phosphorylation. Oncogene. 2002;21:8852–8861. doi: 10.1038/sj.onc.1206048. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
