Synergistic action of microwave-induced mild hyperthermia and paclitaxel in inducing apoptosis in the human breast cancer cell line MCF-7
- PMID: 30655807
- PMCID: PMC6313200
- DOI: 10.3892/ol.2018.9629
Synergistic action of microwave-induced mild hyperthermia and paclitaxel in inducing apoptosis in the human breast cancer cell line MCF-7
Abstract
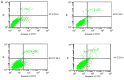
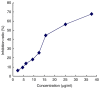
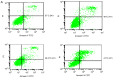
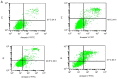
Microwave mild hyperthermia and paclitaxel have been reported to be involved in variety of solid tumors. However, rare related researches have been accomplished via directly killing tumor cells using thermochemotherapy. In order to clarify the potential synergy between microwave-induced hyperthermia at temperatures <41°C and paclitaxel chemotherapy for inhibiting the growth of the human breast cancer cell line MCF-7, an MTT assay was used. The MCF-7 cells cultured in vitro were treated with paclitaxel alone, treated with microwave-induced hyperthermia for 2 h alone (at 40, 40.5 or 41°C), or treated with a combination of paclitaxel and 2 h of hyperthermia (at 40, 40.5 or 41°C). Flow cytometry was used to determine the cell apoptosis rate and it was demonstrated that paclitaxel decreased cell viability in a dose-dependent manner. Alone, hyperthermia for 2 h at 41°C induced apoptosis in MCF-7 cells, to a greater extent compared with hyperthermia for 2 h at 40.0 or 40.5°C (P<0.05). Together, paclitaxel and 2 h of hyperthermia at 40.5°C induced significantly increased apoptosis compared with either treatment alone (P<0.05). Increasing the temperature to 41°C in combination with paclitaxel increased the apoptotic ratio from 12.21±1.02% to 16.36±2.39%. The apoptotic ratio correlated positively with hyperthermia temperature and duration following hyperthermia, as did the synergistic effect obtained by combining hyperthermia and paclitaxel. Notably, the combination of 5 µg/ml paclitaxel and 2 h of hyperthermia at 40°C enhanced MCF-7 cell proliferation. Mild hyperthermia may exert anti-tumor effects by inducing apoptosis, and combining hyperthermia with paclitaxel synergistically induces apoptosis. Paclitaxel dose and hyperthermia temperature require careful optimization, as low-dose paclitaxel combined with hyperthermia at an insufficient temperature may enhance breast cancer proliferation.
Keywords: apoptosis; combined treatment; human breast cancer cell line MCF-7; microwave mild hyperthermia; paclitaxel.
Figures









Similar articles
-
Inhibitory effects of mild hyperthermia plus docetaxel therapy on ER(+/-) breast cancer cells and action mechanisms.J Huazhong Univ Sci Technolog Med Sci. 2013 Dec;33(6):870-876. doi: 10.1007/s11596-013-1214-8. Epub 2013 Dec 13. J Huazhong Univ Sci Technolog Med Sci. 2013. PMID: 24337851
-
Short-term exposure of cancer cells to micromolar doses of paclitaxel, with or without hyperthermia, induces long-term inhibition of cell proliferation and cell death in vitro.Ann Surg Oncol. 2007 Mar;14(3):1220-8. doi: 10.1245/s10434-006-9305-4. Epub 2007 Jan 7. Ann Surg Oncol. 2007. PMID: 17206477
-
Pharmacological and small interference RNA-mediated inhibition of breast cancer-associated fatty acid synthase (oncogenic antigen-519) synergistically enhances Taxol (paclitaxel)-induced cytotoxicity.Int J Cancer. 2005 May 20;115(1):19-35. doi: 10.1002/ijc.20754. Int J Cancer. 2005. PMID: 15657900
-
Short-term hyperthermia promotes the sensitivity of MCF-7 human breast cancer cells to paclitaxel.Biol Pharm Bull. 2013;36(3):376-83. doi: 10.1248/bpb.b12-00774. Epub 2012 Dec 11. Biol Pharm Bull. 2013. PMID: 23229357
-
A moderate thermal dose is sufficient for effective free and TSL based thermochemotherapy.Adv Drug Deliv Rev. 2020;163-164:145-156. doi: 10.1016/j.addr.2020.03.006. Epub 2020 Apr 2. Adv Drug Deliv Rev. 2020. PMID: 32247801 Review.
Cited by
-
Organic dots (O-dots) for theranostic applications: preparation and surface engineering.RSC Adv. 2021 Jan 11;11(4):2253-2291. doi: 10.1039/d0ra08041a. eCollection 2021 Jan 6. RSC Adv. 2021. PMID: 35424170 Free PMC article. Review.
-
Survival of Women with Advanced Stage Cervical Cancer: Neo-Adjuvant Chemotherapy Followed by Radiotherapy and Hyperthermia versus Chemoradiotherapy.Cancers (Basel). 2024 Feb 1;16(3):635. doi: 10.3390/cancers16030635. Cancers (Basel). 2024. PMID: 38339386 Free PMC article.
-
Guidelines for Regulated Cell Death Assays: A Systematic Summary, A Categorical Comparison, A Prospective.Front Cell Dev Biol. 2021 Mar 4;9:634690. doi: 10.3389/fcell.2021.634690. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33748119 Free PMC article. Review.
-
Apoptosis Effects of Oxalis corniculata L. Extract on Human MCF-7 Breast Cancer Cell Line:Galen Med J. 2022 Nov 16;11:e2484. doi: 10.31661/gmj.v11i.2484. eCollection 2022. Galen Med J. 2022. PMID: 36698692 Free PMC article.
-
Hyperthermia Treatment as a Promising Anti-Cancer Strategy: Therapeutic Targets, Perspective Mechanisms and Synergistic Combinations in Experimental Approaches.Antioxidants (Basel). 2022 Mar 24;11(4):625. doi: 10.3390/antiox11040625. Antioxidants (Basel). 2022. PMID: 35453310 Free PMC article. Review.
References
-
- Carrick S, Parker S, Thornton CE, Ghersi D, Simes J, Wilcken N. Single agent versus combination chemotherapy for metastatic breast cancer. Cochrane Database Syst Rev. 2009;CD003372 - PubMed
-
- O'Brien ME, Wigler N, Inbar M, Rosso R, Grischke E, Santoro A, Catane R, Kieback DG, Tomczak P, Ackland SP, et al. Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYX/Doxil) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann Oncol. 2004;15:440–449. doi: 10.1093/annonc/mdh097. - DOI - PubMed
LinkOut - more resources
Full Text Sources
