Sensorimotor plasticity after spinal cord injury: a longitudinal and translational study
- PMID: 30656185
- PMCID: PMC6331953
- DOI: 10.1002/acn3.679
Sensorimotor plasticity after spinal cord injury: a longitudinal and translational study
Abstract
Objective: The objective was to track and compare the progression of neuroplastic changes in a large animal model and humans with spinal cord injury.
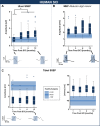
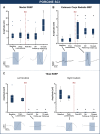
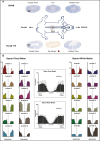
Methods: A total of 37 individuals with acute traumatic spinal cord injury were followed over time (1, 3, 6, and 12 months post-injury) with repeated neurophysiological assessments. Somatosensory and motor evoked potentials were recorded in the upper extremities above the level of injury. In a reverse-translational approach, similar neurophysiological techniques were examined in a porcine model of thoracic spinal cord injury. Twelve Yucatan mini-pigs underwent a contusive spinal cord injury at T10 and tracked with somatosensory and motor evoked potentials assessments in the fore- and hind limbs pre- (baseline, post-laminectomy) and post-injury (10 min, 3 h, 12 weeks).
Results: In both humans and pigs, the sensory responses in the cranial coordinates of upper extremities/forelimbs progressively increased from immediately post-injury to later time points. Motor responses in the forelimbs increased immediately after experimental injury in pigs, remaining elevated at 12 weeks. In humans, motor evoked potentials were significantly higher at 1-month (and remained so at 1 year) compared to normative values.
Conclusions: Despite notable differences between experimental models and the human condition, the brain's response to spinal cord injury is remarkably similar between humans and pigs. Our findings further underscore the utility of this large animal model in translational spinal cord injury research.
Figures


Similar articles
-
Characterization of Motor and Somatosensory Evoked Potentials in the Yucatan Micropig Using Transcranial and Epidural Stimulation.J Neurotrauma. 2017 Sep 15;34(18):2595-2608. doi: 10.1089/neu.2016.4511. Epub 2016 Nov 28. J Neurotrauma. 2017. PMID: 27251314
-
Neurophysiological detection of impending spinal cord injury during scoliosis surgery.J Bone Joint Surg Am. 2007 Nov;89(11):2440-9. doi: 10.2106/JBJS.F.01476. J Bone Joint Surg Am. 2007. PMID: 17974887
-
Sensorimotor training promotes functional recovery and somatosensory cortical map reactivation following cervical spinal cord injury.Eur J Neurosci. 2009 Dec;30(12):2356-67. doi: 10.1111/j.1460-9568.2009.07019.x. Epub 2009 Dec 10. Eur J Neurosci. 2009. PMID: 20092578
-
Pediatric spinal cord injury in infant piglets: description of a new large animal model and review of the literature.J Spinal Cord Med. 2010;33(1):43-57. doi: 10.1080/10790268.2010.11689673. J Spinal Cord Med. 2010. PMID: 20397443 Free PMC article. Review.
-
Afferent input and sensory function after human spinal cord injury.J Neurophysiol. 2018 Jan 1;119(1):134-144. doi: 10.1152/jn.00354.2017. Epub 2017 Jul 12. J Neurophysiol. 2018. PMID: 28701541 Free PMC article. Review.
Cited by
-
Effects of Growth Hormone Treatment and Rehabilitation in Incomplete Chronic Traumatic Spinal Cord Injury: Insight from Proteome Analysis.J Pers Med. 2020 Oct 21;10(4):183. doi: 10.3390/jpm10040183. J Pers Med. 2020. PMID: 33096745 Free PMC article.
-
Porcine Model of Spinal Cord Injury: A Systematic Review.Neurotrauma Rep. 2022 Sep 1;3(1):352-368. doi: 10.1089/neur.2022.0038. eCollection 2022. Neurotrauma Rep. 2022. PMID: 36204385 Free PMC article.
-
Trans-Spinal Electrical Stimulation Therapy for Functional Rehabilitation after Spinal Cord Injury: Review.J Clin Med. 2022 Mar 11;11(6):1550. doi: 10.3390/jcm11061550. J Clin Med. 2022. PMID: 35329875 Free PMC article. Review.
-
Cortical layer-specific modulation of neuronal activity after sensory deprivation due to spinal cord injury.J Physiol. 2021 Oct;599(20):4643-4669. doi: 10.1113/JP281901. Epub 2021 Sep 28. J Physiol. 2021. PMID: 34418097 Free PMC article.
-
Primary motor hand area corticospinal excitability indicates overall functional recovery after spinal cord injury.Front Neurol. 2023 Jun 2;14:1175078. doi: 10.3389/fneur.2023.1175078. eCollection 2023. Front Neurol. 2023. PMID: 37333013 Free PMC article.
References
-
- Hou S, Rabchevsky AG. Autonomic consequences of spinal cord injury. Compr Physiol 2014;4:1419–1453. - PubMed
-
- Kwon BK, Tetzlaff W, Grauer JN, et al. Pathophysiology and pharmacologic treatment of acute spinal cord injury. Spine J 2004;4:451–464. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15246307 - PubMed
-
- Kakulas AB. A review of the neuropathology of human spinal cord injury with emphasis on special features. J Spinal Cord Med 1999;22:119–124. - PubMed
-
- Topka H, Cohen LG, Cole RA, Hallett M. Reorganization of corticospinal pathways following spinal cord injury. Neurology 1991;41:1276–1283. - PubMed
-
- Curt A, Alkadhi H, Crelier GR, et al. Changes of non‐affected upper limb cortical representation in paraplegic patients as assessed by fMRI. Brain 2002;125(Pt 11):2567–2578. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
