Comparing the Self-Assembly of Sexiphenyl-Dicarbonitrile on Graphite and Graphene on Cu(111)
- PMID: 30657213
- PMCID: PMC6519158
- DOI: 10.1002/chem.201806312
Comparing the Self-Assembly of Sexiphenyl-Dicarbonitrile on Graphite and Graphene on Cu(111)
Abstract
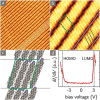
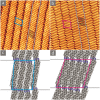
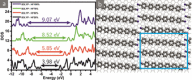
A comparative study on the self-assembly of sexiphenyl-dicarbonitrile on highly oriented pyrolytic graphite and single-layer graphene on Cu(111) is presented. Despite an overall low molecule-substrate interaction, the close-packed structures exhibit a peculiar shift repeating every four to five molecules. This shift has hitherto not been reported for similar systems and is hence a unique feature induced by the graphitic substrates.
Keywords: graphene; nanostructures; scanning probe microscopy; self-assembly; surface analysis.
© 2019 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
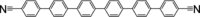
Similar articles
-
1,3,5-Benzenetribenzoic Acid on Cu(111) and Graphene/Cu(111): A Comparative STM Study.J Phys Chem C Nanomater Interfaces. 2016 Aug 18;120(32):18093-18098. doi: 10.1021/acs.jpcc.6b05541. Epub 2016 Jul 20. J Phys Chem C Nanomater Interfaces. 2016. PMID: 27588158 Free PMC article.
-
Structural selection of graphene supramolecular assembly oriented by molecular conformation and alkyl chain.Proc Natl Acad Sci U S A. 2008 Nov 4;105(44):16849-54. doi: 10.1073/pnas.0809427105. Epub 2008 Oct 30. Proc Natl Acad Sci U S A. 2008. PMID: 18974221 Free PMC article.
-
Can graphene be used as a substrate for Raman enhancement?Nano Lett. 2010 Feb 10;10(2):553-61. doi: 10.1021/nl903414x. Nano Lett. 2010. PMID: 20039694
-
Orthogonal Supramolecular Polymer Formation on Highly Oriented Pyrolytic Graphite (HOPG) Surfaces Characterized by Scanning Probe Microscopy.Langmuir. 2015 Oct 27;31(42):11525-31. doi: 10.1021/acs.langmuir.5b02883. Epub 2015 Oct 16. Langmuir. 2015. PMID: 26457462
-
Substrate Effects in the Supramolecular Assembly of 1,3,5-Benzene Tricarboxylic Acid on Graphite and Graphene.Langmuir. 2015 Jun 30;31(25):7016-24. doi: 10.1021/la5048886. Epub 2015 Feb 3. Langmuir. 2015. PMID: 25594568
Cited by
-
Low-Dimensional Metal-Organic Coordination Structures on Graphene.J Phys Chem C Nanomater Interfaces. 2019 May 23;123(20):12730-12735. doi: 10.1021/acs.jpcc.9b00326. Epub 2019 Apr 30. J Phys Chem C Nanomater Interfaces. 2019. PMID: 31156737 Free PMC article.
References
-
- Novoselov K. S., Fal′ko V. I., Colombo L., Gellert P. R., Schwab M. G., Kim K., Nature 2012, 490, 192–200. - PubMed
-
- Ferrari A. C., Bonaccorso F., Fal′ko V., Novoselov K. S., Roche S., Boggild P., Borini S., Koppens F. H. L., Palermo V., Pugno N., Garrido J. A., Sordan R., Bianco A., Ballerini L., Prato M., Lidorikis E., Kivioja J., Marinelli C., Ryhanen T., Morpurgo A., Coleman J. N., Nicolosi V., Colombo L., Fert A., Garcia-Hernandez M., Bachtold A., Schneider G. F., Guinea F., Dekker C., Barbone M., Sun Z., Galiotis C., Grigorenko A. N., Konstantatos G., Kis A., Katsnelson M., Vandersypen L., Loiseau A., Morandi V., Neumaier D., Treossi E., Pellegrini V., Polini M., Tredicucci A., Williams G. M., Hong B. H., Ahn J.-H., Kim J. M., Zirath H., van Wees B. J., van der Zant H., Occhipinti L., Di Matteo A., Kinloch I. A., Seyller T., Quesnel E., Feng X., Teo K., Rupesinghe N., Hakonen P., Neil S. R. T., Tannock Q., Lofwander T., Kinaret J., Nanoscale 2015, 7, 4598–4810. - PubMed
-
- Schmidt N., Stöhr M. in Encycl. Interfacial Chem. Surf. Sci. Electrochem. (Ed.: K. Wandelt), Elsevier, 2018, pp. 110–119.
-
- Mali K. S., Greenwood J., Adisoejoso J., Phillipson R., De Feyter S., Nanoscale 2015, 7, 1566–1585. - PubMed
-
- Macleod J. M., Rosei F., Small 2014, 10, 1038–1049. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

