Tripping the light fantastic in membrane redox biology: linking dynamic structures to function in ER electron transfer chains
- PMID: 30657259
- PMCID: PMC6563164
- DOI: 10.1111/febs.14757
Tripping the light fantastic in membrane redox biology: linking dynamic structures to function in ER electron transfer chains
Abstract
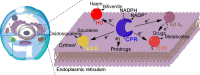
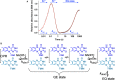
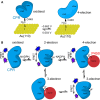
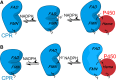
How the dynamics of proteins assist catalysis is a contemporary issue in enzymology. In particular, this holds true for membrane-bound enzymes, where multiple structural, spectroscopic and biochemical approaches are needed to build up a comprehensive picture of how dynamics influence enzyme reaction cycles. Of note are the recent studies of cytochrome P450 reductases (CPR)-P450 (CYP) endoplasmic reticulum redox chains, showing the relationship between dynamics and electron flow through flavin and haem redox centres and the impact this has on monooxygenation chemistry. These studies have led to deeper understanding of mechanisms of electron flow, including the timing and control of electron delivery to protein-bound cofactors needed to facilitate CYP-catalysed reactions. Individual and multiple component systems have been used to capture biochemical behaviour and these have led to the emergence of more integrated models of catalysis. Crucially, the effects of membrane environment and composition on reaction cycle chemistry have also been probed, including effects on coenzyme binding/release, thermodynamic control of electron transfer, conformational coupling between partner proteins and vectorial versus 'off pathway' electron flow. Here, we review these studies and discuss evidence for the emergence of dynamic structural models of electron flow along human microsomal CPR-P450 redox chains.
Keywords: cytochrome P450; cytochrome P450 reductase; electron transfer chemistry; membrane protein; protein domain dynamics.
© 2019 The Authors. The FEBS Journal published by John Wiley & Sons Ltd on behalf of Federation of European Biochemical Societies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Coupled motions direct electrons along human microsomal P450 Chains.PLoS Biol. 2011 Dec;9(12):e1001222. doi: 10.1371/journal.pbio.1001222. Epub 2011 Dec 20. PLoS Biol. 2011. PMID: 22205878 Free PMC article.
-
Real-time analysis of conformational control in electron transfer reactions of human cytochrome P450 reductase with cytochrome c.FEBS J. 2015 Nov;282(22):4357-75. doi: 10.1111/febs.13501. Epub 2015 Sep 16. FEBS J. 2015. PMID: 26307151 Free PMC article.
-
Stopped-flow kinetic studies of electron transfer in the reductase domain of neuronal nitric oxide synthase: re-evaluation of the kinetic mechanism reveals new enzyme intermediates and variation with cytochrome P450 reductase.Biochem J. 2002 Oct 1;367(Pt 1):19-30. doi: 10.1042/BJ20020667. Biochem J. 2002. PMID: 12079493 Free PMC article.
-
[NADPH-cytochrome P450 reductase, not only the partner of cytochrome P450].Postepy Biochem. 2009;55(3):272-8. Postepy Biochem. 2009. PMID: 19928583 Review. Polish.
-
P450BM-3; a tale of two domains--or is it three?Steroids. 1997 Jan;62(1):117-23. doi: 10.1016/s0039-128x(96)00169-9. Steroids. 1997. PMID: 9029725 Review.
Cited by
-
Protein Dynamics and Enzymatic Catalysis.J Phys Chem B. 2023 Mar 30;127(12):2649-2660. doi: 10.1021/acs.jpcb.3c00477. Epub 2023 Mar 21. J Phys Chem B. 2023. PMID: 36944023 Free PMC article. Review.
-
Molecular Lego of Human Cytochrome P450: The Key Role of Heme Domain Flexibility for the Activity of the Chimeric Proteins.Int J Mol Sci. 2022 Mar 25;23(7):3618. doi: 10.3390/ijms23073618. Int J Mol Sci. 2022. PMID: 35408976 Free PMC article.
-
Mechanisms of Hypercapnia-Induced Endoplasmic Reticulum Dysfunction.Front Physiol. 2021 Nov 19;12:735580. doi: 10.3389/fphys.2021.735580. eCollection 2021. Front Physiol. 2021. PMID: 34867444 Free PMC article. Review.
-
A Review: Cytochrome P450 in Alcoholic and Non-Alcoholic Fatty Liver Disease.Diabetes Metab Syndr Obes. 2024 Apr 1;17:1511-1521. doi: 10.2147/DMSO.S449494. eCollection 2024. Diabetes Metab Syndr Obes. 2024. PMID: 38586542 Free PMC article. Review.
-
Unexpected Roles of a Tether Harboring a Tyrosine Gatekeeper Residue in Modular Nitrite Reductase Catalysis.ACS Catal. 2019 Jul 5;9(7):6087-6099. doi: 10.1021/acscatal.9b01266. Epub 2019 May 29. ACS Catal. 2019. PMID: 32051772 Free PMC article.
References
-
- Henzler‐Wildman KA, Lei M, Thai V, Kerns SJ, Karplus M & Kern D (2007) A hierarchy of timescales in protein dynamics is linked to enzyme catalysis. Nature 450, 913–916. - PubMed
-
- Sambongi Y, Iko Y, Tanabe M, Omote H, Iwamoto‐Kihara A, Ueda I, Yanagida T, Wada Y & Futai M (1999) Mechanical rotation of the c subunit oligomer in ATP synthase (F0F1): direct observation. Science 286, 1722–1724. - PubMed
-
- Henzler‐Wildman KA, Thai V, Lei M, Ott M, Wolf‐Watz M, Fenn T, Pozharski E, Wilson MA, Petsko GA, Karplus M et al (2007) Intrinsic motions along an enzymatic reaction trajectory. Nature 450, 838–844. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

