Roles of EphA1/A2 and ephrin-A1 in cancer
- PMID: 30657619
- PMCID: PMC6398892
- DOI: 10.1111/cas.13942
Roles of EphA1/A2 and ephrin-A1 in cancer
Abstract
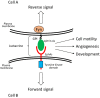
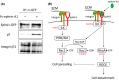
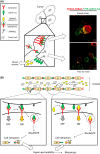
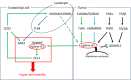
The biological functions of the Eph/ephrin system have been intensively investigated and well documented so far since its discovery in 1987. Although the Eph/ephrin system has been implicated in pathological settings such as Alzheimer's disease and cancer, the molecular mechanism of the Eph/ephrin system in those diseases is not well understood. Especially in cancer, recent studies have demonstrated that most of Eph and ephrin are up- or down-regulated in various types of cancer, and have been implicated in tumor progression, tumor malignancy, and prognosis. However, they lack consistency and are in controversy. The localization patterns of EphA1 and EphA2 in mouse lungs are very similar, and both knockout mice showed similar phenotypes in the lungs. Ephrin-A1 that is a membrane-anchored ligand for EphAs was co-localized with EphA1 and EphA2 in lung vascular endothelial cells. We recently uncovered the molecular mechanism of ephrin-A1-induced lung metastasis by understanding the physiological function of ephrin-A1 in lungs. This review focuses on the function of EphA1, EphA2, and ephrin-A1 in tumors and an establishment of pre-metastatic microenvironment in the lungs.
Keywords: ADAM; Eph; S100A8; ephrin; metastasis.
© 2019 The Authors. Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Conflict of interest statement
The authors have no conflict of interest.
Figures
Similar articles
-
ADAM12-cleaved ephrin-A1 contributes to lung metastasis.Oncogene. 2014 Apr 24;33(17):2179-90. doi: 10.1038/onc.2013.180. Epub 2013 May 20. Oncogene. 2014. PMID: 23686306
-
The Functions of EphA1 Receptor Tyrosine Kinase in Several Tumors.Curr Med Chem. 2023;30(20):2340-2353. doi: 10.2174/0929867329666220820125638. Curr Med Chem. 2023. PMID: 35996244 Review.
-
Eph-A2 promotes permeability and inflammatory responses to bleomycin-induced lung injury.Am J Respir Cell Mol Biol. 2012 Jan;46(1):40-7. doi: 10.1165/rcmb.2011-0044OC. Am J Respir Cell Mol Biol. 2012. PMID: 21799118 Free PMC article.
-
Ephrin-A1 inhibits NSCLC tumor growth via induction of Cdx-2 a tumor suppressor gene.BMC Cancer. 2012 Jul 23;12:309. doi: 10.1186/1471-2407-12-309. BMC Cancer. 2012. PMID: 22824143 Free PMC article.
-
Ephs and ephrins in cancer: ephrin-A1 signalling.Semin Cell Dev Biol. 2012 Feb;23(1):109-15. doi: 10.1016/j.semcdb.2011.10.019. Epub 2011 Oct 25. Semin Cell Dev Biol. 2012. PMID: 22040911 Free PMC article. Review.
Cited by
-
Targeting the EphA2 pathway: could it be the way for bone sarcomas?Cell Commun Signal. 2024 Sep 9;22(1):433. doi: 10.1186/s12964-024-01811-7. Cell Commun Signal. 2024. PMID: 39252029 Free PMC article. Review.
-
Genetically Modified Cellular Therapies for Malignant Gliomas.Int J Mol Sci. 2021 Nov 26;22(23):12810. doi: 10.3390/ijms222312810. Int J Mol Sci. 2021. PMID: 34884607 Free PMC article. Review.
-
Evaluation of Colocasia esculenta Schott in anti-cancerous properties with proximity extension assays.Food Nutr Res. 2021 Oct 4;65. doi: 10.29219/fnr.v65.7549. eCollection 2021. Food Nutr Res. 2021. PMID: 34908921 Free PMC article.
-
EphA1 aggravates neuropathic pain by activating CXCR4/RhoA/ROCK2 pathway in mice.Hum Cell. 2023 Jul;36(4):1416-1428. doi: 10.1007/s13577-023-00911-9. Epub 2023 May 10. Hum Cell. 2023. PMID: 37162645
-
EPHA mutation as a predictor of immunotherapeutic efficacy in lung adenocarcinoma.J Immunother Cancer. 2020 Dec;8(2):e001315. doi: 10.1136/jitc-2020-001315. J Immunother Cancer. 2020. PMID: 33303576 Free PMC article.
References
-
- Hirai H, Maru Y, Hagiwara K, Nishida J, Takaku F. A novel putative tyrosine kinase receptor encoded by the eph gene. Science. 1987;238:1717‐1720. - PubMed
-
- Ieguchi K. Eph as a target in inflammation. Endocr Metab Immune Disord Drug Targets. 2015;15:119‐128. - PubMed
-
- Unified nomenclature for Eph family receptors and their ligands, the ephrins. Eph Nomenclature Committee. Cell. 1997;90:403‐404. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

