Integrated physiological and proteomic analysis of embryo and endosperm reveals central salt stress response proteins during seed germination of winter wheat cultivar Zhengmai 366
- PMID: 30658564
- PMCID: PMC6339335
- DOI: 10.1186/s12870-019-1643-z
Integrated physiological and proteomic analysis of embryo and endosperm reveals central salt stress response proteins during seed germination of winter wheat cultivar Zhengmai 366
Retraction in
-
Retraction Note to: Integrated physiological and proteomic analysis of embryo and endosperm reveals central salt stress response proteins during seed germination of winter wheat cultivar Zhengmai 366.BMC Plant Biol. 2019 Sep 17;19(1):406. doi: 10.1186/s12870-019-2019-0. BMC Plant Biol. 2019. PMID: 31526364 Free PMC article.
Abstract
Background: Salinity is a major abiotic stressor that affects seed germination, plant growth, and crop production. Seed germination represents the beginning of plant growth and is closely linked with subsequent crop development and ultimate yield formation. This study attempted to extend findings regarding the potential proteomic dynamics during wheat seed germination under salt stress and to explore the mechanism of crop salt response.
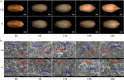
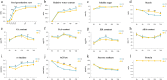
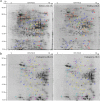
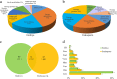
Results: Salt stress significantly affected seed physiological activities during the germination process, resulting in significant decreases in phytohormone and α-amylase activity and significant increases in soluble sugar, starch, and ADP glucose pyrophosphorylase activity. A comparative proteomics approach was applied to analyze the dynamic proteome changes of embryo and endosperm during seed germination in Chinese winter wheat cultivar Zhengmai 366 under salt stress. Two-dimensional electrophoresis identified 92 and 61 differentially accumulated proteins (DAPs) in response to salt stress in embryo and endosperm, respectively. Both organs contained a high proportion of DAPs involved in stress defense, energy metabolism, and protein/amino acid metabolism. The endosperm had more DAPs related to storage proteins and starch metabolism than the embryo, and 2% of DAPs participating in lipid and sterol metabolism were specifically detected in the embryo.
Conclusions: Seed physiological activities were significantly affected during the germination process when subjected to salt stress. The DAPs involved in stress defense and energy metabolism were upregulated whereas those related to reserve substance degradation and protein/amino acid metabolism were significantly downregulated, leading to delayed seed germination under salt stress. Our proteomic results revealed synergistic regulation of the response to salt stress during seed germination.
Keywords: Bread wheat; Embryo; Endosperm; Proteome; Salt stress; Seed germination.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
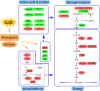
Figures

References
-
- Khan MA, Gulzar S. Germination responses of Sporobolus ioclados: a saline desert grass. J Arid Environ. 2003;53:387–394. doi: 10.1006/jare.2002.1045. - DOI
-
- Rahman S, Kosar-Hashemi B, Samuel MS, Hill A, Abbott DC, Skerritt JH, Morell MK. The major proteins of wheat endosperm starch granules. Funct Plant Biol. 1995;22(5):793–803. doi: 10.1071/PP9950793. - DOI
-
- Wilson JD, Bechtel DB, Todd TC, Seib PA. Measurement of wheat starch granule size distribution using image analysis and laser diffraction technology. Cereal Chem. 2006;83(3):259–268. doi: 10.1094/CC-83-0259. - DOI
-
- Peng M, Gao M, Abdel-Aal ES, Hucl P, Chibbar RN. Separation and characterization of A- and B-type starch granules in wheat endosperm. Cereal Chem. 1999;76(3):375–379. doi: 10.1094/CCHEM.1999.76.3.375. - DOI
-
- Stoddard FL. Genetics of starch granule size distribution in tetraploid and hexaploid wheats. Aust J Agric Res. 2003;54(7):637–648. doi: 10.1071/AR03016. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

