Improved smallest peptides based on positive charge increase of the γ-core motif from Pν D1 and their mechanism of action against Candida species
- PMID: 30666103
- PMCID: PMC6331069
- DOI: 10.2147/IJN.S187957
Improved smallest peptides based on positive charge increase of the γ-core motif from Pν D1 and their mechanism of action against Candida species
Abstract
Background: Plant defensins have a hallmark γ-core motif (GXCX3-9C) that is related to their antimicrobial properties. The aim of this work was to design synthetic peptides based on the region corresponding to the PvD1 defensin γ-core that are the smallest amino acid sequences that bear the strongest biological activity.
Methods: We made rational substitutions of negatively charged amino acid residues with positively charged ones, and the reduction in length in the selected PvD1 γ-core sequence to verify whether the increased net positive charges and shortened length are related to the increase in antifungal activity. Herein, we opted to evaluate the action mechanism of γ33-41 PvD1 ++ peptide due to its significant inhibitory effect on tested yeasts. In addition, it is the smallest construct comprising only nine amino acid residues, giving it a better possibility to be a prototype for designing a new antifungal drug, with lower costs to the pharmaceutical industry while still maintaining the strongest antimicrobial properties.
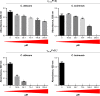
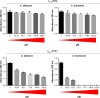
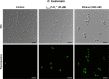
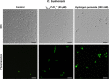
Results: The γ33-41 PvD1 ++ peptide caused the most toxic effects in the yeast Candida buinensis, leading to membrane permeabilization, viability loss, endogenous reactive oxygen species increase, the activation of metacaspase, and the loss of mitochondrial functionality, suggesting that this peptide triggers cell death via apoptosis.
Conclusion: We observed that the antifungal activity of PvD1 is not strictly localized in the structural domain, which comprises the γ-core region and that the increase in the net positive charge is directly related to the increase in antifungal activity.
Keywords: antimicrobial peptide; cationic; defensin; mechanism of action.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures



References
-
- de Coninck B, Cammue BPA, Thevissen K. Modes of antifungal action and in planta functions of plant defensins and defensin-like peptides. Fungal Biol Rev. 2013;26(4):109–120.
-
- Carvalho AO, Gomes VM. Plant defensins – prospects for the biological functions and biotechnological properties. Peptides. 2009;30(5):1007–1020. - PubMed
-
- Jarczak J, Kościuczuk EM, Lisowski P, et al. Defensins: natural component of human innate immunity. Hum Immunol. 2013;74(9):1069–1079. - PubMed
-
- Jha S, Chattoo BB. Expression of a plant defensin in rice confers resistance to fungal phytopathogens. Transgenic Res. 2010;19(3):373–384. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

