Bladder overactivity and afferent hyperexcitability induced by prostate-to-bladder cross-sensitization in rats with prostatic inflammation
- PMID: 30666643
- PMCID: PMC6441933
- DOI: 10.1113/JP277452
Bladder overactivity and afferent hyperexcitability induced by prostate-to-bladder cross-sensitization in rats with prostatic inflammation
Abstract
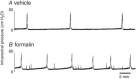
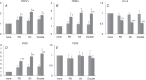
Key points: There is clinical evidence showing that prostatic inflammation contributes to overactive bladder symptoms in male patients; however, little is known about the underlying mechanisms In this study, we investigated the mechanism that prostatic inflammation causes detrusor overactivity by using a rat model of chemically induced prostatic inflammation. We observed a significant number of dorsal root ganglion neurons with dichotomized afferents innervating both prostate and bladder. We also found that prostatic inflammation induces bladder overactivity and urothelial NGF overexpression in the bladder, both dependent on activation of the pelvic nerve, as well as changes in ion channel expression and hyperexcitability of bladder afferent neurons. These results indicate that the prostate-to-bladder cross-sensitization through primary afferent pathways in the pelvic nerve, which contain dichotomized afferents, could be an important mechanism contributing to bladder overactivity and afferent hyperexcitability induced by prostatic inflammation.
Abstract: Prostatic inflammation is reportedly an important factor inducing lower urinary tract symptoms (LUTS) including urinary frequency, urgency and incontinence in patients with benign prostatic hyperplasia (BPH). However, the underlying mechanisms inducing bladder dysfunction after prostatic inflammation are not well clarified. We therefore investigated the effects of prostatic inflammation on bladder activity and afferent function using a rat model of non-bacterial prostatic inflammation. We demonstrated that bladder overactivity, evident as decreased voided volume and shorter intercontraction intervals in cystometry, was observed in rats with prostatic inflammation versus controls. Tissue inflammation, evident as increased myeloperoxidase activity, and IL-1α, IL-1β, and IL-6 levels inside the prostate, but not in the bladder, following intraprostatic formalin injection induced an increase in NGF expression in the bladder urothelium, which depended on activation of the pelvic nerve. A significant proportion (18-19%) of dorsal root ganglion neurons were double labelled by dye tracers injected into either bladder or prostate. In rats with prostatic inflammation, TRPV1, TRPA1 and P2X2 increased, and Kv1.4, a potassium channel α-subunit that can form A-type potassium (KA ) channels, decreased at mRNA levels in bladder afferent and double-labelled neurons vs. non-labelled neurons, and slow KA current density decreased in association with hyperexcitability of these neurons. Collectively, non-bacterial inflammation localized in the prostate induces bladder overactivity and enhances bladder afferent function. Thus, prostate-to-bladder afferent cross-sensitization through primary afferents in the pelvic nerve, which contain dichotomized afferents, could underlie storage LUTS in symptomatic BPH with prostatic inflammation.
Keywords: bladder; cross-sensitization; inflammation; prostate.
© 2019 The Authors. The Journal of Physiology © 2019 The Physiological Society.
Figures







References
-
- Aydin M, Wang HZ, Zhang X, Chua R, Downing K, Melman A & DiSanto ME (2012). Large‐conductance calcium‐activated potassium channel activity, as determined by whole‐cell patch clamp recording, is decreased in urinary bladder smooth muscle cells from male rats with partial urethral obstruction. BJU Int 110, E402–E408. - PubMed
-
- Beppu M, Araki I, Yoshiyama M, Du S, Kobayashi H, Zakoji H & Takeda M (2011). Bladder outlet obstruction induced expression of prostaglandin E2 receptor subtype EP4 in the rat bladder: a possible counteractive mechanism against detrusor overactivity. J Urol 186, 2463–2469. - PubMed
-
- Chen Y, Wu X, Liu J, Tang W, Zhao T & Zhang J (2010). Distribution of convergent afferents innervating bladder and prostate at dorsal root ganglia in rats. Urology 76, 764.e1–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

