Arf GAPs as Regulators of the Actin Cytoskeleton-An Update
- PMID: 30669557
- PMCID: PMC6358971
- DOI: 10.3390/ijms20020442
Arf GAPs as Regulators of the Actin Cytoskeleton-An Update
Abstract
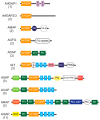
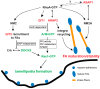
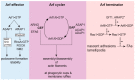
Arf GTPase-activating proteins (Arf GAPs) control the activity of ADP-ribosylation factors (Arfs) by inducing GTP hydrolysis and participate in a diverse array of cellular functions both through mechanisms that are dependent on and independent of their Arf GAP activity. A number of these functions hinge on the remodeling of actin filaments. Accordingly, some of the effects exerted by Arf GAPs involve proteins known to engage in regulation of the actin dynamics and architecture, such as Rho family proteins and nonmuscle myosin 2. Circular dorsal ruffles (CDRs), podosomes, invadopodia, lamellipodia, stress fibers and focal adhesions are among the actin-based structures regulated by Arf GAPs. Arf GAPs are thus important actors in broad functions like adhesion and motility, as well as the specialized functions of bone resorption, neurite outgrowth, and pathogen internalization by immune cells. Arf GAPs, with their multiple protein-protein interactions, membrane-binding domains and sites for post-translational modification, are good candidates for linking the changes in actin to the membrane. The findings discussed depict a family of proteins with a critical role in regulating actin dynamics to enable proper cell function.
Keywords: ADP-ribosylation factor GTPase-activating protein; ADP-ribosylation factors; ARAP1; ARAP2; ASAP1; GIT1/2; actin; focal adhesion; phagocytosis; podosome.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Arf GAPs as regulators of the actin cytoskeleton.Biol Cell. 2007 Oct;99(10):583-600. doi: 10.1042/bc20070034. Biol Cell. 2007. PMID: 17868031 Review.
-
Arf GAPs: A family of proteins with disparate functions that converge on a common structure, the integrin adhesion complex.Small GTPases. 2019 Jul;10(4):280-288. doi: 10.1080/21541248.2017.1299271. Epub 2017 Mar 31. Small GTPases. 2019. PMID: 28362242 Free PMC article. Review.
-
Arf GAPs and molecular motors.Small GTPases. 2019 May;10(3):196-209. doi: 10.1080/21541248.2017.1308850. Epub 2017 Apr 21. Small GTPases. 2019. PMID: 28430047 Free PMC article. Review.
-
Arf GAPs: multifunctional proteins that regulate membrane traffic and actin remodelling.Cell Signal. 2004 Apr;16(4):401-13. doi: 10.1016/j.cellsig.2003.09.012. Cell Signal. 2004. PMID: 14709330 Review.
-
AGAP1, an endosome-associated, phosphoinositide-dependent ADP-ribosylation factor GTPase-activating protein that affects actin cytoskeleton.J Biol Chem. 2002 Dec 13;277(50):48965-75. doi: 10.1074/jbc.M202969200. Epub 2002 Oct 17. J Biol Chem. 2002. PMID: 12388557
Cited by
-
The Drosophila melanogaster Rab GAP RN-tre cross-talks with the Rho1 signaling pathway to regulate nonmuscle myosin II localization and function.Mol Biol Cell. 2020 Oct 1;31(21):2379-2397. doi: 10.1091/mbc.E20-03-0181. Epub 2020 Aug 20. Mol Biol Cell. 2020. PMID: 32816624 Free PMC article.
-
Phosphoinositide signaling at the cytoskeleton in the regulation of cell dynamics.Cell Death Dis. 2025 Apr 14;16(1):296. doi: 10.1038/s41419-025-07616-x. Cell Death Dis. 2025. PMID: 40229242 Free PMC article. Review.
-
Polymorphisms in the ASAP1 and SP110 Genes and Its Association with the Susceptibility to Pulmonary Tuberculosis in a Mongolian Population.J Immunol Res. 2022 Sep 20;2022:2713869. doi: 10.1155/2022/2713869. eCollection 2022. J Immunol Res. 2022. PMID: 36249417 Free PMC article.
-
Polygenic risk for traumatic loss-related PTSD in US military veterans: Protective effect of secure attachment style.World J Biol Psychiatry. 2021 Dec;22(10):792-799. doi: 10.1080/15622975.2021.1907721. Epub 2021 May 20. World J Biol Psychiatry. 2021. PMID: 33821766 Free PMC article.
-
Crystal Structure of the SH3 Domain of ASAP1 in Complex with the Proline Rich Motif (PRM) of MICAL1 Reveals a Unique SH3/PRM Interaction Mode.Int J Mol Sci. 2023 Jan 11;24(2):1414. doi: 10.3390/ijms24021414. Int J Mol Sci. 2023. PMID: 36674928 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

