Location-Dependent Patient Outcome and Recurrence Patterns in IDH1-Wildtype Glioblastoma
- PMID: 30669568
- PMCID: PMC6356480
- DOI: 10.3390/cancers11010122
Location-Dependent Patient Outcome and Recurrence Patterns in IDH1-Wildtype Glioblastoma
Abstract
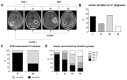
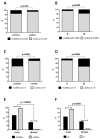
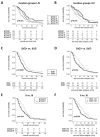
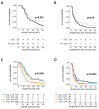
Recent studies suggest that glioblastomas (GBMs) contacting the subventricular zone (SVZ) as the main adult neurogenic niche confer a dismal prognosis but disregard the unique molecular and prognostic phenotype associated with isocitrate dehydrogenase 1 (IDH1) mutations. We therefore examined location-dependent prognostic factors, growth, and recurrence patterns in a consecutive cohort of 285 IDH1-wildtype GBMs. Based on pre-operative contrast-enhanced MRI, patients were allotted to four location-dependent groups with (SVZ+; groups I, II) and without (SVZ-; groups III, IV) SVZ involvement or with (cortex+; groups I, III) and without (cortex-; groups II, IV) cortical involvement and compared for demographic, treatment, imaging, and survival data at first diagnosis and recurrence. SVZ involvement was associated with lower Karnofsky performance score (p < 0.001), lower frequency of complete resections at first diagnosis (p < 0.0001), and lower non-surgical treatment intensity at recurrence (p < 0.001). Multivariate survival analysis employing a Cox proportional hazards model identified SVZ involvement as an independent prognosticator of inferior overall survival (p < 0.001) and survival after relapse (p = 0.041). In contrast, multifocal growth at first diagnosis (p = 0.031) and recurrence (p < 0.001), as well as distant recurrences (p < 0.0001), was more frequent in cortex+ GBMs. These findings offer the prospect for location-tailored prognostication and treatment based on factors assessable on pre-operative MRI.
Keywords: Glioblastoma; distant recurrence; isocitrate dehydrogenase 1 (IDH1)-wildtype; multifocal growth; subventricular zone; survival.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Spatial transcriptome analysis reveals Notch pathway-associated prognostic markers in IDH1 wild-type glioblastoma involving the subventricular zone.BMC Med. 2016 Oct 26;14(1):170. doi: 10.1186/s12916-016-0710-7. BMC Med. 2016. PMID: 27782828 Free PMC article.
-
Relationship of glioblastoma multiforme to neural stem cell regions predicts invasive and multifocal tumor phenotype.Neuro Oncol. 2007 Oct;9(4):424-9. doi: 10.1215/15228517-2007-023. Epub 2007 Jul 10. Neuro Oncol. 2007. PMID: 17622647 Free PMC article.
-
The influence of subventricular zone involvement in extent of resection and tumor growth pattern of glioblastoma.Innov Surg Sci. 2020 Aug 27;5(3-4):127-132. doi: 10.1515/iss-2020-0011. eCollection 2020 Sep. Innov Surg Sci. 2020. PMID: 34966832 Free PMC article.
-
Risk Factors of Distant Recurrence and Dissemination of IDH Wild-Type Glioblastoma: A Single-Center Study and Meta-Analysis.Cancers (Basel). 2024 Aug 18;16(16):2873. doi: 10.3390/cancers16162873. Cancers (Basel). 2024. PMID: 39199644 Free PMC article. Review.
-
[Isocitrate dehydrogenase type I mutation as a prognostic factor in glioblastoma and a literature review].Neurocirugia (Astur). 2015 Nov-Dec;26(6):276-83. doi: 10.1016/j.neucir.2015.04.001. Epub 2015 Jul 18. Neurocirugia (Astur). 2015. PMID: 26194445 Review. Spanish.
Cited by
-
WHERE DID THE TUMOR START? AN INVERSE SOLVER WITH SPARSE LOCALIZATION FOR TUMOR GROWTH MODELS.Inverse Probl. 2020 Apr;36(4):045006. doi: 10.1088/1361-6420/ab649c. Epub 2020 Feb 26. Inverse Probl. 2020. PMID: 33746330 Free PMC article.
-
Prognostic analysis and nomogram construction for older patients with IDH-wild-type glioblastoma.Heliyon. 2023 Jul 17;9(7):e18310. doi: 10.1016/j.heliyon.2023.e18310. eCollection 2023 Jul. Heliyon. 2023. PMID: 37519736 Free PMC article.
-
Risk Estimation in Non-Enhancing Glioma: Introducing a Clinical Score.Cancers (Basel). 2023 Apr 27;15(9):2503. doi: 10.3390/cancers15092503. Cancers (Basel). 2023. PMID: 37173969 Free PMC article.
-
Recurrent Glioblastoma: From Molecular Landscape to New Treatment Perspectives.Cancers (Basel). 2020 Dec 26;13(1):47. doi: 10.3390/cancers13010047. Cancers (Basel). 2020. PMID: 33375286 Free PMC article. Review.
-
Fluorescence Guidance in Glioma Surgery: A Narrative Review of Current Evidence and the Drive Towards Objective Margin Differentiation.Cancers (Basel). 2025 Jun 17;17(12):2019. doi: 10.3390/cancers17122019. Cancers (Basel). 2025. PMID: 40563668 Free PMC article. Review.
References
-
- Sanai N., Tramontin A.D., Quinones-Hinojosa A., Barbaro N.M., Gupta N., Kunwar S., Lawton M.T., McDermott M.W., Parsa A.T., Verdugo J.M., et al. Unique astrocyte ribbon in adult human brain contains neural stem cells but lacks chain migration. Nature. 2004;427:740–744. doi: 10.1038/nature02301. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

