Evaluation of automated malaria diagnosis using the Sysmex XN-30 analyser in a clinical setting
- PMID: 30670023
- PMCID: PMC6341646
- DOI: 10.1186/s12936-019-2655-8
Evaluation of automated malaria diagnosis using the Sysmex XN-30 analyser in a clinical setting
Abstract
Background: Early and accurate diagnosis of malaria is a critical aspect of efforts to control the disease, and several diagnostic tools are available. Microscopic assessment of a peripheral blood smear enables direct visualization of parasites in infected red blood cells and is the clinical diagnostic gold standard. However, it is subjective and requires a high level of skill. Numerous indirect detection methods are in use, but are not ideal since surrogate markers of infection are measured. This study describes the first clinical performance evaluation of the automated Sysmex XN-30 analyser, which utilizes fluorescence flow cytometry to directly detect and quantitate parasite-infected red blood cells.
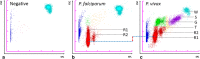
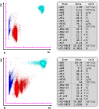
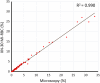
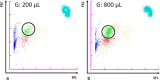
Results: Residual EDTA blood samples from suspected malaria cases referred for routine diagnosis were analysed on the XN-30. Parasitaemia was reported as a percentage, as well as absolute numbers of infected red blood cells, and scattergrams provided a visual image of the parasitized red blood cell clusters. The results reported by the XN-30 correlated with microscopy and the analyser demonstrated 100% sensitivity and specificity. Measurements were reproducible and storage of samples at room temperature did not affect the parameters. Several Plasmodium species were detected, including Plasmodium falciparum, Plasmodium vivax and Plasmodium ovale. The XN-30 also identified the transmissible gametocytes as a separate cluster on the scattergrams. Abnormal red blood cell indices (low haemoglobin and raised reticulocyte counts), haemoglobinopathies and thrombocytopenia did not interfere with the detection of parasites. The XN-30 also generated a concurrent full blood count for each sample.
Conclusions: The novel technology of the Sysmex XN-30 provides a robust, rapid, automated and accurate platform for diagnosing malaria in a clinical setting. The objective enumeration of red blood cells infected with Plasmodium species makes it suitable for global use and allows monitoring of the parasite load once therapy has been initiated, thereby providing an early marker of drug resistance. The automated generation of a full blood count for each sample provides an opportunity for detecting unsuspected cases. Asymptomatic carriers can also be identified, which will be useful in blood transfusion centres, and will enable treatment of these individuals to prevent the spread of the disease.
Keywords: Automated diagnosis; Plasmodium falciparum; Sysmex XN-30 analyser.
Figures


Similar articles
-
An automated haematology analyzer XN-30 distinguishes developmental stages of falciparum malaria parasite cultured in vitro.Malar J. 2018 Feb 2;17(1):59. doi: 10.1186/s12936-018-2208-6. Malar J. 2018. PMID: 29391022 Free PMC article.
-
The XN-30 hematology analyzer for rapid sensitive detection of malaria: a diagnostic accuracy study.BMC Med. 2019 May 31;17(1):103. doi: 10.1186/s12916-019-1334-5. BMC Med. 2019. PMID: 31146732 Free PMC article. Clinical Trial.
-
Automated Plasmodium detection by the Sysmex XN hematology analyzer.J Clin Pathol. 2018 Jul;71(7):594-599. doi: 10.1136/jclinpath-2017-204878. Epub 2018 Jan 3. J Clin Pathol. 2018. PMID: 29298814
-
Computational microscopic imaging for malaria parasite detection: a systematic review.J Microsc. 2015 Oct;260(1):1-19. doi: 10.1111/jmi.12270. Epub 2015 Jun 5. J Microsc. 2015. PMID: 26047029
-
Misidentification of Plasmodium ovale as Plasmodium vivax malaria by a microscopic method: a meta-analysis of confirmed P. ovale cases.Sci Rep. 2020 Dec 11;10(1):21807. doi: 10.1038/s41598-020-78691-7. Sci Rep. 2020. PMID: 33311528 Free PMC article.
Cited by
-
Selective Hole Filling of Red Blood Cells for Improved Marker-Controlled Watershed Segmentation.Scanning. 2021 Dec 6;2021:5678117. doi: 10.1155/2021/5678117. eCollection 2021. Scanning. 2021. PMID: 34950282 Free PMC article.
-
Rapid Detection of Plasmodium vivax by the Hematology Analyzer for Population Screening.Diagnostics (Basel). 2023 Nov 7;13(22):3397. doi: 10.3390/diagnostics13223397. Diagnostics (Basel). 2023. PMID: 37998533 Free PMC article.
-
White blood cell differential fluorescence abnormal scattergram: A useful indicator for early detection of malarial parasite.Pak J Med Sci. 2022 Mar-Apr;38(3Part-I):687-691. doi: 10.12669/pjms.38.3.4702. Pak J Med Sci. 2022. PMID: 35480534 Free PMC article.
-
Diagnostic accuracy of fluorescence flow-cytometry technology using Sysmex XN-31 for imported malaria in a non-endemic setting.Parasite. 2022;29:31. doi: 10.1051/parasite/2022031. Epub 2022 May 31. Parasite. 2022. PMID: 35638753 Free PMC article.
-
Fast detection and quantification of Plasmodium species infected erythrocytes in a non-endemic region by using the Sysmex XN-31 analyzer.Malar J. 2022 Apr 11;21(1):119. doi: 10.1186/s12936-022-04147-0. Malar J. 2022. PMID: 35410230 Free PMC article.
References
-
- WHO. World malaria report 2018. Geneva: World Health Organization; 2018. http://www.who.int/malaria/publications/world-malaria-report-2018/en/. Accessed 22 Nov 2018.
-
- Chotivanich K, Silamut K, Day N. Laboratory diagnosis of malaria infection—a short review of methods. N Z J Med Lab Sci. 2007;61:4–7.
-
- WHO. Guidelines for the treatment of malaria. 3rd edition. Geneva: World Health Organization; 2015. http://www.who.int/malaria/publications/atoz/9789241549127/en/. Accessed 14 Nov 2018.
-
- Hopkins H, Gonzalez IJ, Polley SD, Angutoko P, Ategeka J, Asiimwe C, et al. Highly sensitive detection of malaria parasitemia in a malaria-endemic setting: performance of a new loop-mediated isothermal amplification kit in a remote clinic in Uganda. J Infect Dis. 2013;208:645–652. doi: 10.1093/infdis/jit184. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

