Hypoxia Mimetic Agents for Ischemic Stroke
- PMID: 30671433
- PMCID: PMC6331394
- DOI: 10.3389/fcell.2018.00175
Hypoxia Mimetic Agents for Ischemic Stroke
Abstract
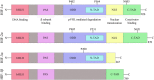
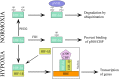
Every year stroke claims more than 6 million lives worldwide. The majority of them are ischemic stroke. Small molecule-based therapeutics for ischemic stroke has attracted a lot of attention, but none has been shown to be clinically useful so far. Hypoxia-inducible factor-1 (HIF-1) plays a crucial role in the transcriptional adaptation of cells to hypoxia. Small molecule-based hypoxia-mimetic agents either stabilize HIF-1α via HIF-prolyl hydroxylases (PHDs) inhibition or through other mechanisms. In both the cases, these agents have been shown to confer ischemic neuroprotection in vitro and in vivo. The agents which act via PHD inhibition are mainly classified into iron chelators, iron competitors, and 2 oxoglutarate (2OG) analogs. This review discusses HIF structure and key players in the HIF-1 degradation pathway as well as the genes, proteins and chemical molecules that are connected to HIF-1 and how they affect cell survival following ischemic injury. Furthermore, this review gives a summary of studies that used PHD inhibitors and other HIF-1α stabilizers as hypoxia-mimetic agents for the treatment of ischemic injury.
Keywords: hypoxia mimetic agent; hypoxia-inducible factor-1; iron chelators; ischemic stroke; neuroprotection.
Figures
References
-
- Baranova O., Miranda L. F., Pichiule P., Dragatsis I., Johnson R. S., Chavez J. C. (2007). Neuron-specific inactivation of the hypoxia inducible factor 1 increases brain injury in a mouse model of transient focal cerebral ischemia. J. Neurosci. 27 6320–6332. 10.1523/jneurosci.0449-07.2007 - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources