Comparison of in vitro static and dynamic assays to evaluate the efficacy of an antimicrobial drug combination against Staphylococcus aureus
- PMID: 30673774
- PMCID: PMC6344103
- DOI: 10.1371/journal.pone.0211214
Comparison of in vitro static and dynamic assays to evaluate the efficacy of an antimicrobial drug combination against Staphylococcus aureus
Abstract
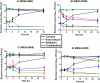
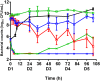
An easily implementable strategy to reduce treatment failures in severe bacterial infections is to combine already available antibiotics. However, most in vitro combination assays are performed by exposing standard bacterial inocula to constant concentrations of antibiotics over less than 24h, which can be poorly representative of clinical situations. The aim of this study was to assess the ability of static and dynamic in vitro Time-Kill Studies (TKS) to identify the potential benefits of an antibiotic combination (here, amikacin and vancomycin) on two different inoculum sizes of two S. aureus strains. In the static TKS (sTKS), performed by exposing both strains over 24h to constant antibiotic concentrations, the activity of the two drugs combined was not significantly different the better drug used alone. However, the dynamic TKS (dTKS) performed over 5 days by exposing one strain to fluctuating concentrations representative of those observed in patients showed that, with the large inoculum, the activities of the drugs, used alone or in combination, significantly differed over time. Vancomycin did not kill bacteria, amikacin led to bacterial regrowth whereas the combination progressively decreased the bacterial load. Thus, dTKS revealed an enhanced effect of the combination on a large inoculum not observed in sTKS. The discrepancy between the sTKS and dTKS results highlights that the assessment of the efficacy of a combination for severe infections associated with a high bacterial load could be demanding. These situations probably require the implementation of dynamic assays over the entire expected treatment duration rather than the sole static assays performed with steady drug concentrations over 24h.
Conflict of interest statement
The authors have the following interests: DCB, FEG and FW are employed by Vetoquinol SA. There are no patents, products in development or marketed products to declare. This does not alter the authors’ adherence to all the PLOS ONE policies on sharing data and materials.
Figures
Similar articles
-
In vitro evaluation of antibiotics' combinations for empirical therapy of suspected methicillin resistant Staphylococcus aureus severe respiratory infections.BMC Infect Dis. 2007 Sep 21;7:111. doi: 10.1186/1471-2334-7-111. BMC Infect Dis. 2007. PMID: 17888153 Free PMC article.
-
In vitro activity effects of combinations of cephalothin, dicloxacillin, imipenem, vancomycin and amikacin against methicillin-resistant Staphylococcus spp. strains.Ann Clin Microbiol Antimicrob. 2006 Oct 12;5:25. doi: 10.1186/1476-0711-5-25. Ann Clin Microbiol Antimicrob. 2006. PMID: 17034644 Free PMC article.
-
Novel Antibiotic-loaded Point-of-care Implant Coating Inhibits Biofilm.Clin Orthop Relat Res. 2015 Jul;473(7):2270-82. doi: 10.1007/s11999-014-4130-8. Clin Orthop Relat Res. 2015. PMID: 25604874 Free PMC article.
-
Vancomycin and piperacillin-tazobactam against methicillin-resistant Staphylococcus aureus and vancomycin-intermediate Staphylococcus aureus in an in vitro pharmacokinetic/pharmacodynamic model.Clin Ther. 2014 Oct 1;36(10):1334-44. doi: 10.1016/j.clinthera.2014.06.027. Epub 2014 Jul 25. Clin Ther. 2014. PMID: 25066667
-
Differential Activity of the Combination of Vancomycin and Amikacin on Planktonic vs. Biofilm-Growing Staphylococcus aureus Bacteria in a Hollow Fiber Infection Model.Front Microbiol. 2018 Mar 27;9:572. doi: 10.3389/fmicb.2018.00572. eCollection 2018. Front Microbiol. 2018. PMID: 29636741 Free PMC article.
Cited by
-
Exploring the Antimicrobial and Pharmacological Potential of NF22 as a Potent Inhibitor of E. coli DNA Gyrase: An In Vitro and In Silico Study.Pharmaceutics. 2022 Dec 10;14(12):2768. doi: 10.3390/pharmaceutics14122768. Pharmaceutics. 2022. PMID: 36559262 Free PMC article.
-
Effects of Incubation Time and Inoculation Level on the Stabilities of Bacteriostatic and Bactericidal Antibiotics against Salmonella Typhimurium.Antibiotics (Basel). 2021 Aug 22;10(8):1019. doi: 10.3390/antibiotics10081019. Antibiotics (Basel). 2021. PMID: 34439069 Free PMC article.
-
Pharmacodynamic Functions of Synthetic Derivatives for Treatment of Methicillin-Resistant Staphylococcus aureus (MRSA) and Mycobacterium tuberculosis.Front Microbiol. 2020 Nov 27;11:551189. doi: 10.3389/fmicb.2020.551189. eCollection 2020. Front Microbiol. 2020. PMID: 33329419 Free PMC article.
-
Dynamic In Vitro PK/PD Infection Models for the Development and Optimisation of Antimicrobial Regimens: A Narrative Review.Antibiotics (Basel). 2024 Dec 10;13(12):1201. doi: 10.3390/antibiotics13121201. Antibiotics (Basel). 2024. PMID: 39766591 Free PMC article. Review.
-
Effect of Ciprofloxacin, Levofloxacin, and Ofloxacin on Pseudomonas aeruginosa: A case control study with time kill curve analysis.Ann Med Surg (Lond). 2022 Sep 16;82:104674. doi: 10.1016/j.amsu.2022.104674. eCollection 2022 Oct. Ann Med Surg (Lond). 2022. PMID: 36268299 Free PMC article.
References
-
- Clinical and Laboratory Standards Institute. CLSI. Methods for Dilution Antimicrobial Susceptibility Tests f or Bacteria That Grow Aerobically; Approved Standard—Ninth Edition. CLSI document M07-A9. Clinical and Laboratory Standards Institute; 2012.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

