Chiral Assembly Preferences and Directing Effects in Supramolecular Two-Component Organogels
- PMID: 30674807
- PMCID: PMC6209267
- DOI: 10.3390/gels4020031
Chiral Assembly Preferences and Directing Effects in Supramolecular Two-Component Organogels
Abstract
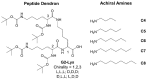
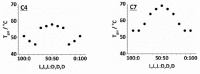
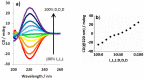
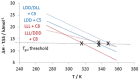
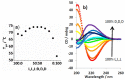
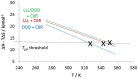
The impact of chirality on the self-assembly of supramolecular gels is of considerable importance, as molecular-scale programming can be translated into nanostructuring and ultimately affect macroscopic performance. This paper explores the effect of chirality on the assembly of two-component gels comprised of a second-generation dendritic lysine peptide acid, containing three chiral centres, and an amine. This combination forms an acid⁻amine complex that assembles into nanofibres through peptide-peptide hydrogen bonds, leading to organogels. With achiral amines, a racemic mixture of l,l,l and d,d,d dendritic peptide acids surprisingly forms the best gels-more commonly, mixing enantiomers suppresses gelation. Thermodynamic studies demonstrate that depending on the amine, the greater stability of heterochiral gels can either be entropically or enthalpically driven. With amines possessing "R" chirality, the l,l,l peptide acid consistently forms more effective gels than its d,d,d analogue. Furthermore, in mixed gels, l,l,l sometimes imposes its assembly preference onto d,d,d. In summary, this paper demonstrates a rare example in which heterochiral gels are preferred, and also explores directing effects when each component in a two-component gel is chiral.
Keywords: chirality; organogel; peptide; self-assembly; two-component.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Diffusion across a gel-gel interface - molecular-scale mobility of self-assembled 'solid-like' gel nanofibres in multi-component supramolecular organogels.Chem Sci. 2018 May 30;9(25):5541-5550. doi: 10.1039/c8sc01071d. eCollection 2018 Jul 7. Chem Sci. 2018. PMID: 30061985 Free PMC article.
-
Two-component dendritic gel: effect of stereochemistry on the supramolecular chiral assembly.Chemistry. 2004 Nov 19;10(23):5901-10. doi: 10.1002/chem.200400502. Chemistry. 2004. PMID: 15472938
-
Effect of Stereochemistry on Chirality and Gelation Properties of Supramolecular Self-Assemblies.Chemistry. 2021 Feb 10;27(9):3119-3129. doi: 10.1002/chem.202004533. Epub 2021 Jan 15. Chemistry. 2021. PMID: 33225542
-
Entropically Favoured Assembly of Pyrazine-Based Helical Fibers into Superstructures: Achiral/ Chiral Guest-Induced Chirality Transformation.Angew Chem Int Ed Engl. 2019 Nov 4;58(45):16203-16209. doi: 10.1002/anie.201908669. Epub 2019 Sep 25. Angew Chem Int Ed Engl. 2019. PMID: 31502340 Review.
-
Enantioselective Recognition by Chiral Supramolecular Gels.Chem Asian J. 2016 Oct 6;11(19):2642-2649. doi: 10.1002/asia.201600441. Epub 2016 Jun 3. Chem Asian J. 2016. PMID: 27258582 Review.
Cited by
-
Supramolecular Fractal Growth of Self-Assembled Fibrillar Networks.Gels. 2021 Apr 14;7(2):46. doi: 10.3390/gels7020046. Gels. 2021. PMID: 33919860 Free PMC article.
-
Using chirality to influence supramolecular gelation.Chem Sci. 2019 Jul 3;10(33):7801-7806. doi: 10.1039/c9sc02239b. eCollection 2019 Sep 7. Chem Sci. 2019. PMID: 31588329 Free PMC article.
-
Diffusion across a gel-gel interface - molecular-scale mobility of self-assembled 'solid-like' gel nanofibres in multi-component supramolecular organogels.Chem Sci. 2018 May 30;9(25):5541-5550. doi: 10.1039/c8sc01071d. eCollection 2018 Jul 7. Chem Sci. 2018. PMID: 30061985 Free PMC article.
-
Understanding multicomponent low molecular weight gels from gelators to networks.J Adv Res. 2025 Mar;69:91-106. doi: 10.1016/j.jare.2024.03.028. Epub 2024 Apr 1. J Adv Res. 2025. PMID: 38570015 Free PMC article. Review.
References
-
- Banerjee S., Das R.K., Maitra U. Supramolecular Gels ‘In Action’. J. Mater. Chem. 2009;19:6649–6687. doi: 10.1039/b819218a. - DOI
LinkOut - more resources
Full Text Sources

